|
 |
- Search
| J Stroke > Volume 26(2); 2024 > Article |
|
Abstract
Distal medium vessel occlusions (DMVOs) are thought to cause as many as 25% to 40% of all acute ischemic strokes and may result in substantial disability amongst survivors. Although intravenous thrombolysis (IVT) is more effective for distal than proximal vessel occlusions, the overall efficacy of IVT remains limited in DMVO with less than 50% of patients achieving reperfusion and about 1/3 to 1/4 of the patients failing to achieve functional independence. Data regarding mechanical thrombectomy (MT) among these patients remains limited. The smaller, thinner, and more tortuous vessels involved in DMVO are presumably associated with higher procedural risks whereas a lower benefit might be expected given the smaller amount of tissue territory at risk. Recent advances in technology have shown promising results in endovascular treatment of DMVOs with room for future improvement. In this review, we discuss some of the key technical and clinical considerations in DMVO treatment including the anatomical and clinical terminology, diagnostic modalities, the role of IVT and MT, existing technology, and technical challenges as well as the contemporary evidence and future treatment directions.
Distal medium vessel occlusions (DMVOs) account for approximately 25% to 40% of all acute ischemic strokes (AIS) [1,2]. Despite their relatively high frequency, there is no consensus regarding the optimal management of these patients. The current AHA/ASA (American Stroke Association/American Heart Association) guidelines state that “Although the benefits are uncertain, the use of mechanical thrombectomy (MT) with stent retrievers may be reasonable for carefully selected patients with AIS in whom treatment can be initiated (groin puncture) within 6 hours of symptom onset and who have causative occlusion of the M2 or M3 segments of the middle cerebral artery (MCA) as well as of the anterior cerebral arteries (ACA), vertebral arteries, basilar artery, or posterior cerebral arteries (PCA) (level IIb).” [3] The ESO/ESMINT (European Stroke Organisation-European Society for Minimally Invasive Neurological Therapy) guidelines concur that “patients with M2 occlusion fulfilled the inclusion criteria in most randomized trials and therefore MT is reasonable in this situation” but does not make any specific recommendation regarding endovascular treatment (EVT) for occlusions involving the M3 segments of the MCA, the ACA, or the PCA [4]. However, the fact that AIS related to DMVO often results in significant disability despite best medical treatment including intravenous thrombolysis (IVT) calls for novel treatment approaches [5,6].
Data regarding MT in DMVO stroke patients remains relatively limited. Among the recent EVT randomized trials demonstrating EVT efficacy, DMVO was found in a small minority of the patients with most of the “distal occlusions” involving a dominant (supplying >50% of the territory, n=73) or co-dominant (supplying 50% of the territory, n=50) MCA-M2 [7,8]. In fact, there were only seven non-dominant M2 occlusions in the expanded HERMES meta-analysis and “other occlusions” comprised only 2% (n=23) of all patients included in the initial HERMES meta-analysis encompassing 1,287 patients across the first five randomized EVT trials [7]. Fortunately, a growing number of non-randomized studies have now been published demonstrating the feasibility of EVT for DMVO strokes [1,9-13]. These studies have demonstrated that distal EVT leads to high rates of successful reperfusion and may be performed with a comparable safety profile to that of EVT for proximal arterial occlusions. However, non-randomized studies are known to be associated with high risk of biases. Therefore, there is a strong rationale to test the safety and efficacy of EVT for DMVO strokes in prospective randomized clinical trials. Our aim is to provide an objective review of the literature on the topic of DMVO including a brief discussion about the ongoing and planned clinical trials.
There have been recent attempts to better define the nomenclature for the different vascular territories involved in AIS. Saver et al. [2] proposed that cerebrovascular occlusions should be categorized both in terms of their distance/tortuosity (distal vs. proximal vessel occlusions) and vessel size (large, medium, and small vessel occlusions) with “medium vessels” being operationally defined as cerebral arteries with lumen diameters between 0.75 mm and 2.0 mm. As such, the term proximal large vessel occlusion (PLVO) should be typically applied for those occlusions involving the intracranial internal carotid artery (ICA), the M1 segment of the MCA, the vertebral, and basilar arteries while the term DMVO has been proposed for involvement of the M3-M4, A2-A5, P2-P5, posterior inferior cerebellar artery, anterior inferior cerebellar artery, and superior cerebellar artery. The authors also suggested a variation in the nomenclature for the occlusions involving some of the more proximal vessels such as the MCA-M2, ACA-A1, PCA-P1 depending on their caliber-distal large or distal medium vessel occlusions.
Goyal and Collegues [1,7,14] proposed a different system that from an anatomic standpoint focused mostly on the vessel size with little or no emphasis on the distance/tortuosity component while also incorporating clinical deficits to its definition. The authors suggested the term medium vessel occlusion (MeVO) to describe the usual vessel sizes that allows for safe endovascular thrombectomy based on available technology and techniques, with typical caliber ranging from 1 mm to 3 mm. In order to justify the potential risks associated with MT, a clinical deficit criterion was added to the definition to only include occlusions resulting in National Institutes of Health Stroke Scale (NIHSS) ≥5 or NIHSS <5 with disabling deficits [1,7,14]. We tend to agree with Saver et al. [2] in that vessels between 2 mm and 3 mm should be preferentially considered large rather than medium vessels. A second concern we have about the proposed MeVO definition is that the incorporation of clinical symptoms to an anatomic definition can be problematic. For instance, the term large vessel occlusion (LVO) is well-established and is often utilized even when it is associated with mild or even absent clinical deficits.
Finally, it is important to acknowledge that DMVOs or MeVOs may have primary (de novo occlusions) or secondary presentations. Secondary presentations result from thrombus fragmentation and migration leading to emboli to new territories or to distal territories which may occur spontaneously or after IVT and/or EVT.
Noninvasive imaging at presentation is crucial to establish DMVO/MeVO diagnosis and to assist the treatment decision-making (Figures 1-3). On non-contrast computed tomography, distal rich-in-erythrocyte occlusive thrombi are expected to show a hyperdense “dot” sign in the Sylvian triangle vessels [15]. However, systematic evaluation for dot signs is not well incorporated in clinical practice. Future implementation of artificial intelligence approaches may help overcome some of these challenges. In addition, the hyperdense “dot” sign is highly dependent on clot burden and composition and is not as well reported for ACA, PCA, and cerebellar branch arteries. Likewise, on susceptibility-weighted sequences, magnetic resonance imaging (MRI) can show susceptibility vessel signs (peripheral blooming artifact). This is demonstrated as a low-intensity signal for distal occlusive rich-in-erythrocyte thrombi [16]. In contrast, erythrocyte-poor thrombi are typically not easily visualized. However, they might be indirectly identified via the local slow flow supported by fluid attenuation inversion recovery (FLAIR) vascular hyperintensity [2]. Computed tomography angiography (CTA) and magnetic resonance angiography (MRA) may provide high reliability in detecting MCA-M2, ACA-A1, PCA-P1, and proximal cerebellar artery occlusions. However, confusion can occur in more distal branches due to decreased spatial resolution in face of the reduced artery diameter and the variability in branch anatomy. Hence, it might be challenging to determine whether the loss of distal vessel signal should be explained by an occlusion or anatomic variation. Novel CTA and MRA techniques (waveletCTA and 7-T MRA) may overcome some of these limitations but they have limited availability [17-19]. Therefore, computed tomography perfusion (CTP) can serve as a reliable adjunct imaging tool for DMVO in which occlusion of the feeding vessel is suggested by hypoperfusion in a wedge-shaped region matching the typical territory of distal ACA, MCA, or PCA [20-22]. Interestingly, a recent study assessed the possible role of CTP in stroke imaging protocol for admission in which hypoperfusion areas for vessel occlusion were defined by Tmax >6 seconds, along with the clinical symptoms of the corresponding vascular territory. The false negative rate of vessel occlusion on CTA for patients undergoing EVT was 30.1% (median Tmax >6 s: 69 [46-99.5] mL) which supports the role of CTP in increasing both vessel occlusion diagnosis and EVT rates [23]. The variability of imaging protocols for DMVOs among published observational studies necessitates more research to establish rigorous and specific imaging selection criteria. It is noteworthy that direct to angiosuite (DTAS) is an emerging approach that may improve time metrics and outcomes compared with the direct transfer to conventional imaging (DTCI) [24]. Additionally, DTAS approach for NIHSS ≥10 might also increase the diagnostic frequency and treatment of DMVO with high clinical severity [25].
The cumulative data suggests that, although IVT is comparatively more effective for distal than proximal arterial occlusions, the overall efficacy of alteplase remains limited in the setting of DMVO/MeVO with less than 50% of patients achieving recanalization.
Indeed, partial or complete recanalization with intravenous (IV) alteplase alone was achieved in only 52% of M2-M3 MCA occlusions evaluated in a meta-analysis that included 2,063 patients across 26 studies [26]. Similarly, IV alteplase before EVT recanalized only 43% of MCA-M3, ACA, or PCA and 37% of MCA-M2 occlusions, compared with 22% for MCA-M1 and 11% of ICA occlusions, enrolled in the INTERRSeCT study [27]. In a combined analysis of the INTERRSeCT and PRoveIT studies comprising 258 MeVO (M2/M3/A2/A3/P2/P3 occlusions) patients, early recanalization on follow-up CTA was observed in 47% of patients receiving versus 21% of those not receiving alteplase (P=0.003) [5].
Similarly, the INSPIRE registry (n=945) demonstrated that the post-alteplase IVT rates of complete recanalization on follow-up CTA were higher in DMVO (174 M2, 87 M3, 28 ACA, 43 PCA occlusions) than M1 occlusions (66% vs. 49%, P=0.005) [28]. Notably, the rates of complete reperfusion on follow-up CTP imaging (e.g., perfusion lesion reduction >80% from baseline to 24-hour CTP, or 24-hour perfusion lesion of zero) did not significantly differ across groups (47% vs. 40%, P=0.365) in this study.
The natural history of DMVO/MeVO is highly variable depending on the size and eloquence of the territory supplied by the involved distal artery. In this setting, it becomes reasonable to postulate that the degree of disability rather than the level of arterial occlusion should ideally guide decisions around reperfusion therapy. In fact, there is a good rationale for this as a previous study focusing on ICA, M1, and M2 occlusions concluded that stroke clinical severity (e.g., NIHSS) and collateral flow appear to be more important than the level of proximal intracranial arterial occlusion in determining outcomes [29].
In the INTERRSeCT and PRoveIT combined analysis, amongst the patients treated versus not treated with IVT, there was a significant association of IV alteplase and overall disability (ordinal 90-day modified Rankin Scale [mRS]: adjusted common odds ratio [cOR], 0.55; 95% confidence interval [CI], 0.33-0.92; P=0.044) but not with either excellent (90-day mRS 0-1: 53.2% vs. 41.7%; adjusted OR [aOR], 1.70 [95% CI, 0.88-3.25]) or good (90-day mRS 0-2: 68.3% vs. 65.3%; aOR, 1.54 [95% CI, 0.70-3.36]) clinical outcomes [5]. Successful early recanalization had a robust association with excellent outcome (aOR, 2.29 [95% CI, 1.23-4.28]) and ordinal mRS (adjusted cOR, 0.57 [95% CI, 0.34-0.96]) as well a strong trend towards an association with good outcome (aOR, 2.10 [95% CI, 0.998-4.43]). The overall results remained similar after excluding patients with proximal M2 occlusions from the analysis.
Although functional outcomes in the INSPIRE registry were better in patients with DMVO as compared to M1 occlusions (mRS 0-1: 55% vs. 37%; mRS 0-2: 73% vs. 50%, P<0.001), nearly one in two DMVO patients failed to achieve excellent outcomes and one in four failed to achieve independence [28]. This study also demonstrated that baseline ischemic core was a more powerful predictor of functional outcome than the site of occlusion and that the relationship between ischemic core and outcome did not differ according to site of occlusion.
A meta-analysis of the EXTEND, ECASS-4, and EPITHET trials evaluated the potential benefit of IV alteplase in CTP-selected patients treated between 4.5-9 hours [6]. There were 155 non-LVO patients with functional independence (mRS 0-2) being achieved in 61% of the placebo and 65% of the alteplase patients. A total of 83 non-LVO patients were “mismatch positive” with 64% of the placebo and 68% of the alteplase patients achieving functional independence (BV Campbell, personal communication, 2020). Likewise, there was no significant benefit for IV alteplase either in terms of 90-day mRS 0-1 (OR: 1.44 [95% CI, 0.52-3.96]) or 90-day mRS ordinal shift (OR: 1.65 [95% CI, 0.76-3.61]) even in the presence of mismatch.
Over the last few years, the techniques and technologies of distal EVT have greatly advanced resulting in an increasing body of literature to support the feasibility and safety of EVT for DMVO strokes [1,9-13].
The Trevo Retriever Registry was an international, multicenter, prospective, open-label registry that recruited a total of 2,008 patients at 76 sites across 12 countries between November 2013 and May 2017. There were 407 DMVOs in the study including 350 (86.0%) M2, 25 (6.1%) M3, 10 (2.5%) ACA, and 22 (5.4%) PCA occlusions, out of which 376 had pre-morbid mRS 0-2. These patients were compared with 1,268 PLVO patients[ 1].2 The median baseline NIHSS score was lower in DMVO (13 [8-18] vs. 16 [12-20], P<0.001) but there were no differences in terms of age, sex, IV tissue plasminogen activator use, co-morbidities, or time to treatment. Safety data including the rates of post-procedure reperfusion, symptomatic intracranial hemorrhage (sICH), and 90-mortality were comparable between both groups. DMVO showed significantly higher rates of 90-day mRS 0-2 (68.3% vs. 56.5%, P<0.001). After adjustment for potential confounders, the level of arterial occlusion was not associated with the chances of excellent outcome (DMVO for 90-day mRS 0-1: OR, 1.18 [95% CI, 0.90-1.54], P=0.225), successful reperfusion, or sICH. However, patients with DMVO were more likely to be functionally independent (mRS 0-2: OR, 1.45 [95% CI, 1.09-1.92], P=0.01) at 90 days. Surprisingly, they also had higher mortality within 90 days (OR, 1.54 [95% CI, 1.06-2.27], P=0.02).
An analysis of the STAR Registry including 189 patients with DMVO demonstrated that, as compared to PLVO, DMVO had higher rate of good outcome (90-day mRS 0-2: 45% vs. 36%; P=0.03) and lower rate of successful reperfusion (Thrombolysis in Cerebral Infarction [TICI] 2b/3: 78% vs. 84%; P=0.04) [30]. However, there were no significant differences after adjustments were made for potential confounders. Notably, the rates of hemorrhagic complication (6% vs. 5%, P=0.64) and 90-day mortality (22% vs. 19%, P=0.46) were also similar. Successful reperfusion (TICI 2b/3: OR, 4.7 [95% CI, 1.2-18.6], P=0.026), age (OR, 0.96 [95% CI, 0.92-0.99], P=0.019), and baseline NIHSS scores (OR, 0.91 [95% CI, 0.84-0.98], P=0.016) but not technical modality (contact aspiration [CA] vs. stent retriever [SR] vs. combined therapy as first-line) were identified as predictors of good outcome in the DMVO population.
In another published study from EVATRISP prospective registry, 92 patients with isolated ACA-stroke were identified, 55 (60%) were treated with IVT only and 37 (40%) with EVT (±bridging IVT). Although odds for favorable outcome, sICH and mortality did not differ significantly between both groups, patients treated with EVT tended to have higher stroke severity on admission (NIHSS: 10.0 vs. 7.0, P=0.054) [31]. In a systematic review including nine studies (a total of 168 patients) with MT-treated ACA occlusions, recanalization modified TICI 2b/3 was achieved in 80%, complications occurred in 17%, and mortality at 90 days was 19% [32].
Another multicenter case-control study (TOPMOST study) included 184 matched patients treated for primary distal occlusion of P2 (81%) or P3 (19%). At discharge, the mean difference of NIHSS score decrease between MT group and standard medical management group was -1.5 points (95% CI, 3.2 to -0.8; P=0.06). Interestingly, significant MT results were observed in the subgroup with NIHSS ≥10 (mean difference, -5.6; 95% CI, -10.9 to -0.2; P=0.04) and in the subgroup without IVT (mean difference, -3.0; 95% CI, -5.0 to -0.9; P=0.005). sICH occurred in 4.3% in each treatment cohort [33]. In a sub-analysis of TOPMOST, the results showed that the initial reperfusion approach was aspiration only in 29% and SR in 71%, with similar first-pass effect rates of 53.7% and 44% (P=0.297), respectively. There were also no significant differences in early neurological improvement (aspiration: 64.7% vs. SR: 52.2%; P=0.933) and mRS rates (mRS 0-1, aspiration: 60.5% vs. SR 68.6%; P=0.4) [34].
In a recently published multicenter cohort study, 286 patients with DMVO in anterior circulation were evaluated with 156 treated with EVT and 130 treated with medical managment (MM). There was no difference in the unadjusted rate of 3-month functional independence in the EVT versus MM groups (151 [51.7%] vs. 124 [50.0%]; P=0.78), excellent outcome (151 [38.4%] vs. 123 [31.7%]; P=0.25), or mortality (139 [18.7%] vs. 106 [11.3%]; P=0.15). The rate of sICH was similar in the EVT versus MM groups (weighted: 4.0% vs. 3.1%; P=0.90). In inverse probability of treatment weighting propensity analyses, there was no significant difference between groups for functional independence (aOR, 1.36; 95% CI, 0.84-2.19; P=0.20) or mortality (aOR, 1.24; 95% CI, 0.63- 2.43; P=0.53), whereas the EVT group had higher odds of an excellent outcome (mRS scores, 0-1) at 3 months (aOR, 1.71; 95% CI, 1.02-2.87; P=0.04) [35].
One of the main challenges to the broad implementation of distal thrombectomy is related to the presumable less favorable risk-benefit balance as compared to LVOs. Fortunately, there is emerging data regarding safety of EVT in the distal territory employing newer techniques and devices [1,2].
Smaller devices have been introduced and have shown promising results including the 3 mm Trevo NXT ProVue (Stryker Neurovascular, Fremont, CA, USA), Solitaire X (Medtronic, Minneapolis, MN, USA), Catch Mini (Balt, Montmorency, France), and pREset LITE (phenox GmbH, Bochum, Germany) as well as the adjustable 2.5 mm Tigertriever 13 (Rapid Medical, Yoqneam, Israel). An early experience report of eight cases described the Trevo XP 3×20 mm retriever (Baby Trevo) for DMVO and showed promising results of feasibility in those cases. However, significant vasospasm after the device pass was noted in almost half of the patients but all responded to infusion of an intra-arterial (IA) vasodilator (milrinone and/or nicardipine) [11]. Further studies validated this early experience and built more supporting evidence for Trevo XP 3×20 mm retriever for DMVOs [9,36,37]. Additionally, low profile thrombectomy Tigertriever 13 device showed high rate of successful recanalization and a reasonable safety profile [38]. Notably, nimodipine was systematically used in the flushing solution of the guiding catheter to prevent vasospasm in this study. Likewise, thromboaspiration devices are evolving. Low profile aspiration catheters like 3MAX (Penumbra, Alameda, CA, USA) and Zoom 35 (Imperative Care, Campbell, CA, USA) are designed to be more compatible with the distal vasculature by providing more flexibility, while maintaining appropriate suction force. These devices are highly navigable, and we have had favorable experiences even with trans-circulation approaches [9,36,37].
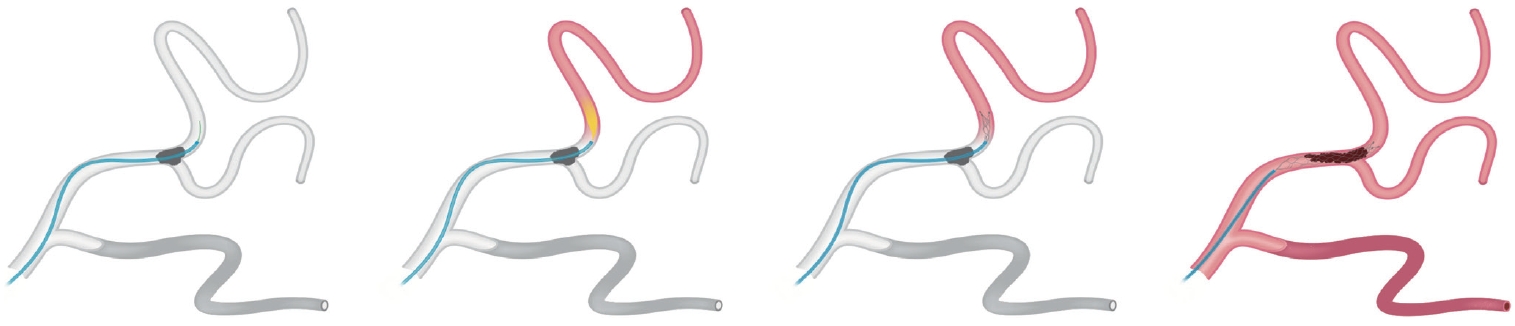
The shorter clot length often found in DMVO makes CA an attractive option. However, one must acknowledge the potential risks of blindly advancing a relatively large diameter device into a vascular territory that typically curves, branches, and tappers as the device sizing may be apparent adequate just proximally to the occlusion but may become dangerously large just past to it. This may lead to significant vascular injury and hemorrhagic complications (Figure 4). In contrast, the use of small SR might be safer as the clot is crossed with a lower profile microcatheter and the SR device will more easily accommodate any unexpected differences in vessel caliber that might occur. Importantly and even more so in distal territories, deployment should be achieved by unsheathing rather than pushing the SR and this should be preferably performed in a relatively straight vessel segment. SRs have the advantage of working as a “probe” of the distal vasculature which may help with the planning of the next steps (Figure 5). However, the SR removal carries the risk of vascular distortion due to both “straightening” and “stretching” effects that may result in the detachment of the small penetrating cortical feeders and hemorrhage (Figure 6). This vascular distortion may be lessened by the placement of an aspiration catheter in the proximal aspect of the SR to provide distal support and optimize the force vector (Figure 7).
In a comparative analysis of 3MAX aspiration versus 3 mm Trevo Retriever, the 3 mm Trevo showed higher rates of first-pass reperfusion than direct 3MAX thromboaspiration [36]. However, the optimal treatment modality choice might be influenced by clot characteristics. In LVOs, clots that are associated with “hyperdense sign” on CT or “blooming artifact” on MRI seem to respond better to SR whereas those that are not associated with these imaging signs may respond better to CA [39,40]. The combined SR and CA approach might be particularly advantageous in the setting of DMVO. In the blind exchange with mini-pinning technique (BeMP), a 3 mm retriever is initially deployed which then provides an “anchor” for the removal of the microcatheter. A small-bore aspiration catheter is then “blindly” advanced over the bare SR delivery wire (blind exchange). Once the SR delivery wire comes out of the aspiration catheter, a gentle pull force can be applied to the wire to facilitate the navigation of the catheter until clot contact is achieved and aspiration applied. The retriever is then partially resheathed in order to “cork” the thrombus (mini-pinning) and finally the system is pulled as a unit [10]. This technique has been shown to be superior to either SR or CA in isolation [13].
There are other critical anatomic factors to consider in the decision-making process. It is typically challenging to perform EVT in the distal superior division branches of the MCA due to their greater tortuosity and multiple branching points. Intra-arterial thrombolytics (IAT) might be a better option in this territory [41]. Deploying an SR to transiently increase the interaction surface for IAT followed by resheathing the SR might be another approach to consider in order to avoid the potential complications associated with the straightening and stretching of the vascular anatomy. Many studies have reported the benefits of IAT as an upfront or rescue strategy with promising beneficial results. In a study that included 112 patients with a hyperdense MCA sign, IAT was more beneficial than IVT with more favorable outcomes after IAT (53%) than after IVT (23%; P=0.001), and lower mortality after IAT (7%) than after IVT (23%; P=0.022) [42]. Similarly, according to the recent CHOICE trial, use of adjunct IAT with alteplase resulted in a greater likelihood of excellent neurological outcome compared with placebo [43]. Finally, general anesthesia in DMVOs EVT might offer a greater advantage compared to moderate sedation as it minimizes patients’ movements during the procedure and subsequently yield to a safer technical course while attempting to catheterize the more distal and fragile target arteries.
Given the lack of solid evidence from randomized controlled trials to support EVT for DMVO, there is a clinical equipoise in DMVO/MeVO treatment. In an international cross-sectional survey exploring the preference of 366 participants from 44 countries, most physicians (59%) would treat patients with DMVOs immediately with EVT. Some of the factors influencing this decision-making were baseline NIHSS, core volume, IV thrombolytic use, patients’ age, and occlusion site. Most physicians were also willing to accept patients transferred for EVT from a primary center (82%) and the majority of these (76.5%) were willing to randomize these patients after transfer [44]. Patient age >65 years, A3 occlusion, small core volume, and patient IV alteplase eligibility significantly influenced the physician’s decision to randomize (aOR 1.24, 95% CI 1.13-1.36; aOR 1.17, 95% CI 1.01-1.34; aOR 0.98, 95% CI 0.97-0.99; and aOR 1.38, 95% CI 1.21-1.57, respectively) [45].
Multicenter randomized controlled trials have been recently proposed to investigate the safety and efficacy of EVT, IAT, and IVT for DMVO, with some few interesting variations in design and intervention (Table 1).
In DISTAL trial (Switzerland, NCT05029414), intervention can be initiated within 6 hours of last known well (LKW) or within 6 to 24 hours of LKW and imaging mismatch criteria on CT or MRI and there is no restriction on the technique and devices following local protocols and physicians’ preference. In DISTALS trial (NCT05152524), Tigertriever 13 device is used in patients with NIHSS 4-24, or NIHSS 2-24 for patients with aphasia and/or hemianopia. The study necessiates perfusion lesion (Tmax >4.0 s) volume ≥10 mL on CTP or MR perfusion-weighted imaging (PWI) within the territory and occluded distal vessel diameter ≥1.5 mm as measured on CTA or MRA. Additionally, ischemic core lesion (relative cerebral blood flow <30% on CTP or apparent diffusion coefficient <620 on MR DWI) in ≤50% of the perfusion lesion volume within 24 hours of LKW is required. The ESCAPE-MeVO trial (NCT05151172) requires the use of the Solitaire X device (Medtronic) within 12 hours. In the French DISCOUNT trial (NCT05030142), the intervention is restricted to 6 hours with NIHSS <5. In DUSK trial (NCT05983757), we are recruiting patients who are ineligible for or have failed IV thrombolytic treatment with primary DMVOs resulting in significant clinical deficits within 12 hours of LKW.
Intravenous thrombolytics are also being considered for DMVO treatment within 4.5-12 hours from time last seen well. The RESILIENT EXTEND-IV (NCT05199662) is a phase III, randomized, multicenter clinical trial in Brazil that aims to assess the role of IV TNK in patients who suffer a non-LVO ischemic stroke within 4.5-12 hours from LKW. Patients are expected to be ineligible for IV thrombolytic treatment with alteplase due to onset >4.5 hours and for EVT under standard of care due to absence of proximal arterial occlusion with evidence of salvageable brain tissue on perfusion imaging.
IAT is under investigation treatment option for incomplete reperfusion and residual occlusions including DMVOs. TECNO (NCT05499832) is a multicenter, randomized trial evaluating if additional administration of IA TNK improves reperfusion in patients with an initial LVO in the anterior circulation with incomplete reperfusion and residual occlusions. For this purpose, 156 adult participants with incomplete reperfusions and residual occlusions after MT are expected to receive either 3 mg IA TNK or medical treatment alone. Per inclusion criteria, onset to randomization should be within 345 minutes from LKW.
Despite the advances in the devices, there are remaining technological limitations that must be addressed. In a case-based international survey among neurointerventionalists, there was an overall belief that there was a substantial room for improvement in tools used for DMVO (68%). In particular, potential advancement can be focused on smaller size stent retrievers (32%), softer device (23%) with less radial force (4%) and more navigable aspiration catheters (31%), longer length (12%) with high aspiration force (6%) [46]. Notably, mini-stentrievers with 1.5 mm and 2 mm diameters are now available in Japan. High heterogeneities regarding the type of DMVO (primary vs. secondary), characteristics of each vessel and anatomical variabilities are one of the most challenging parts of establishing generalizable evidence, managing DMVOs and deciding the optimal treatment approach. We believe there are hierarchical elements that should be factored-in while triaging DMVOs with tortuosity towards the occluded vessel being the most crucial factor followed by the vessel caliber and finally the distance to the occluded vessel. Eloquence mapping can be of additional value to stroke imaging tools before decision to pursue EVT in DMVO patients.
In the absence of high-quality data, it becomes critical to acknowledge that DMVOs occur in smaller, thinner, and more tortuous vessels and as such are potentially associated with a higher treatment risk. DMVOs are also associated with smaller volumes of tissue territory at risk, thus it is likely that treatment will result in lower (if any) benefit. However, despite the best available medical treatments, outcomes are far from ideal. Therefore, EVT becomes a very reasonable option for territories of high eloquence that are associated with severe disability, particularly in the setting of a favorable anatomy and in the hands of experienced operators. As treatment remains largely unproven, it is essential to discuss the potential risks and benefits of treatment even more carefully with the patients and their families.
In summary, DMVO strokes are common representing 25%-40% of all AIS. IVT remains the only proven treatment for DMVOs but results in only modest rates of successful recanalization with significant residual disability in 25%-50% of the patients. Moreover, many patients are not eligible for IVT due to presentation time or other contra-indications. Randomized trials of EVT for PLVO stroke have demonstrated an overwhelming benefit over standard medical treatment (including IVT) and the current data suggests that EVT can be safely performed in the distal cerebrovasculature with technical outcomes that mirror the PLVO experience. This landscape strongly supports the need for randomized clinical trials to properly assess the safety and efficacy of EVT in the treatment of DMVO strokes. If the results of such a trial are positive, international guidelines and systems of care would need to be updated in order to ensure optimal treatment to many patients who otherwise would face severe long-term disability.
Notes
Conflicts of interest
RGN reports consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Philips, Hybernia, Imperative Care, Medtronic, Phenox, Philips, Prolong Pharmaceuticals, Stryker Neurovascular, Shanghai Wallaby, Synchron, and stock options for advisory roles with Astrocyte, Brainomix, Cerebrotech, Ceretrieve, Corindus Vascular Robotics, Vesalio, Viz-AI, RapidPulse, and Perfuze. RGN is one of the principal investigators of the “Endovascular Therapy for Low NIHSS Ischemic Strokes (ENDOLOW)” trial. Funding for this project is provided by Cerenovus. RGN is the principal investigator of the “Combined Thrombectomy for Distal MediUm Vessel Occlusion StroKe (DUSK)” trial. Funding for this project is provided by Stryker Neurovascular. RGN is an investor in Viz-AI, Perfuze, Cerebrotech, Reist/Q’Apel Medical, Truvic, Vastrax, and Viseon. BAG reports consulting fees from Medtronic, Microvention, and Stryker. ARA is a consultant for Cerenovus and Stryker Neurovascular. MR reports grant funding (NIH P30 AG2482 and NIH 5U24 NS107216-05). DCH reports consulting fees for Stryker Neurovascular, Cerenovus, Chiesi USA, Brainomix, Poseydon Medical; Consulting/DSMB: Jacobs Institute/Medtronic, Vesalio; stock options: VizAI. All other authors report no conflict of interest.
Figure 1.
A 39-year-old female with acute onset of right hemiplegia, left gaze, and aphasia. Non-contrast computed tomography demonstrated the presence of hyperdense “dot signs” in the Sylvian (indicated by arrows, A) and opercular (indicated by arrows, B) regions consistent thrombi involving the M2- and M3 segments of the left middle cerebral artery (indicated by arrows, C) which were successfully recanalized post-thrombectomy (D).

Figure 2.
A 68-year-old male with acute onset of left leg weakness. Quantitative computed tomography (CT) perfusion demonstrated a large area of hypoperfusion (Tmax >6 s) involving the right anterior cerebral artery territory with a significant mismatch (A). Conventional angiography confirmed complete occlusion of the A2 segment of the left anterior cerebral artery (indicated by arrows, B) which was successfully recanalized post-thrombectomy (indicated by arrows, C). Post-treatment brain magnetic resonance imaging disclosed an infarct similar to the “ischemic core” (relative CBF <30%) area previously identified on the CT perfusion (D). CBF, cerebral blood flow.

Figure 3.
A 55-year-old male with acute onset of left homonymous hemianopsia. Brain magnetic resonance imaging (MRI) gradient echo sequence showed a “susceptibility vessel sign” consistent with a red-blood-cell-rich thrombus within the distal right posterior cerebral artery (indicated by arrows, A). Qualitative MRI perfusion demonstrated a large area involving the right posterior cerebral artery territory (as circled, B). Conventional angiography confirmed occlusion of the fetal-type distal right posterior cerebral artery (indicated by arrows, C) which was successfully recanalized post-thrombectomy (indicated by arrows, D).

Table 1.
Summary of the ongoing randomized clinical trials for distal medium vessel occlusion
NIHSS, National Institutes of Health Stroke Scale; CTA, computed tomography angiography; MRA, magnetic resonance angiography; CTP, computed tomography perfusion; PWI, perfusion-weighted imaging; PCA, posterior cerebral artery; ACA, anterior cerebral artery; MCA, middle cerebral artery; LKW, last known well.
References
1. Ospel JM, Goyal M. A review of endovascular treatment for medium vessel occlusion stroke. J Neurointerv Surg 2021;13:623-630.


2. Saver JL, Chapot R, Agid R, Hassan A, Jadhav AP, Liebeskind DS, et al. Thrombectomy for distal, medium vessel occlusions: a consensus statement on present knowledge and promising directions. Stroke 2020;51:2872-2884.


3. Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2019;50:e344-e418.

4. Turc G, Bhogal P, Fischer U, Khatri P, Lobotesis K, Mazighi M, et al. European Stroke Organisation (ESO) - European Society for Minimally Invasive Neurological Therapy (ESMINT) guidelines on mechanical thrombectomy in acute ischaemic strokeendorsed by Stroke Alliance for Europe (SAFE). Eur Stroke J 2019;4:6-12.




5. Ospel JM, Menon BK, Demchuk AM, Almekhlafi MA, Kashani N, Mayank A, et al. Clinical course of acute ischemic stroke due to medium vessel occlusion with and without intravenous alteplase treatment. Stroke 2020;51:3232-3240.


6. Campbell BCV, Ma H, Ringleb PA, Parsons MW, Churilov L, Bendszus M, et al. Extending thrombolysis to 4·5-9 h and wake-up stroke using perfusion imaging: a systematic review and meta-analysis of individual patient data. Lancet 2019;394:139-147.

7. Goyal M, Menon BK, van Zwam WH, Dippel DW, Mitchell PJ, Demchuk AM, et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet 2016;387:1723-1731.


8. Menon BK, Hill MD, Davalos A, Roos YBWEM, Campbell BCV, Dippel DWJ, et al. Efficacy of endovascular thrombectomy in patients with M2 segment middle cerebral artery occlusions: meta-analysis of data from the HERMES collaboration. J Neurointerv Surg 2019;11:1065-1069.


9. Grossberg JA, Rebello LC, Haussen DC, Bouslama M, Bowen M, Barreira CM, et al. Beyond large vessel occlusion strokes: distal occlusion thrombectomy. Stroke 2018;49:1662-1668.


10. Haussen DC, Al-Bayati AR, Eby B, Ravindran K, Rodrigues GM, Frankel MR, et al. Blind exchange with mini-pinning technique for distal occlusion thrombectomy. J Neurointerv Surg 2020;12:392-395.


11. Haussen DC, Lima A, Nogueira RG. The Trevo XP 3×20 mm retriever (‘Baby Trevo’) for the treatment of distal intracranial occlusions. J Neurointerv Surg 2016;8:295-299.


12. Nogueira RG, Mohammaden MH, Haussen DC, Budzik RF, Gupta R, Krajina A, et al. Endovascular therapy in the distal neurovascular territory: results of a large prospective registry. J Neurointerv Surg 2021;13:979-984.


13. Pérez-García C, Moreu M, Rosati S, Simal P, Egido JA, GomezEscalonilla C, et al. Mechanical thrombectomy in medium vessel occlusions: blind exchange with mini-pinning technique versus mini stent retriever alone. Stroke 2020;51:3224-3231.


14. Goyal M, Ospel JM, Menon BK, Hill MD. MeVO: the next frontier? J Neurointerv Surg 2020;12:545-547.


15. Leary MC, Kidwell CS, Villablanca JP, Starkman S, Jahan R, Duckwiler GR, et al. Validation of computed tomographic middle cerebral artery “dot” sign: an angiographic correlation study. Stroke 2003;34:2636-2640.


16. Park MG, Oh SJ, Baik SK, Jung DS, Park KP. Susceptibility-weighted imaging for detection of thrombus in acute cardioembolic stroke. J Stroke 2016;18:73-79.




17. Kunz WG, Sommer WH, Havla L, Dorn F, Meinel FG, Dietrich O, et al. Detection of single-phase CTA occult vessel occlusions in acute ischemic stroke using CT perfusion-based wavelettransformed angiography. Eur Radiol 2017;27:2657-2664.



18. Mattern H, Sciarra A, Godenschweger F, Stucht D, Lüsebrink F, Rose G, et al. Prospective motion correction enables highest resolution time-of-flight angiography at 7T. Magn Reson Med 2018;80:248-258.




19. Park CA, Kang CK, Kim YB, Cho ZH. Advances in MR angiography with 7T MRI: from microvascular imaging to functional angiography. Neuroimage 2018;168:269-278.


20. Amukotuwa SA, Wu A, Zhou K, Page I, Brotchie P, Bammer R. Distal medium vessel occlusions can be accurately and rapidly detected using Tmax maps. Stroke 2021;52:3308-3317.


21. Becks MJ, Manniesing R, Vister J, Pegge SAH, Steens SCA, van Dijk EJ, et al. Brain CT perfusion improves intracranial vessel occlusion detection on CT angiography. J Neuroradiol 2019;46:124-129.


22. Sousa JA, Sondermann A, Bernardo-Castro S, Varela R, Donato H, Sargento-Freitas J. CTA and CTP for detecting distal medium vessel occlusions: a systematic review and meta-analysis. AJNR Am J Neuroradiol 2023;45:51-56.


23. Olive-Gadea M, Requena M, Diaz F, Boned S, Garcia-Tornel A, Muchada M, et al. Systematic CT perfusion acquisition in acute stroke increases vascular occlusion detection and thrombectomy rates. J Neurointerv Surg 2022;14:1270-1273.


24. Mohammaden MH, Doheim MF, Elfil M, Al-Bayati AR, Pinheiro A, Nguyen TN, et al. Direct to angiosuite versus conventional imaging in suspected large vessel occlusion: a systemic review and meta-analysis. Stroke 2022;53:2478-2487.


25. Requena M, Olivé-Gadea M, Muchada M, Hernández D, Rubiera M, Boned S, et al. Direct to angiography suite without stopping for computed tomography imaging for patients with acute stroke: a randomized clinical trial. JAMA Neurol 2021;78:1099-1107.



26. Seners P, Turc G, Maïer B, Mas JL, Oppenheim C, Baron JC. Incidence and predictors of early recanalization after intravenous thrombolysis: a systematic review and meta-analysis. Stroke 2016;47:2409-2412.


27. Menon BK, Al-Ajlan FS, Najm M, Puig J, Castellanos M, Dowlatshahi D, et al. Association of clinical, imaging, and thrombus characteristics with recanalization of visible intracranial occlusion in patients with acute ischemic stroke. JAMA 2018;320:1017-1026.



28. Tian H, Parsons MW, Levi CR, Lin L, Aviv RI, Spratt NJ, et al. Influence of occlusion site and baseline ischemic core on outcome in patients with ischemic stroke. Neurology 2019;92:e2626-e2643.


29. Lima FO, Furie KL, Silva GS, Lev MH, Camargo EC, Singhal AB, et al. Prognosis of untreated strokes due to anterior circulation proximal intracranial arterial occlusions detected by use of computed tomography angiography. JAMA Neurol 2014;71:151-157.


30. Anadani M, Alawieh A, Chalhoub R, Jabbour P, Starke RM, Arthur A, et al. Mechanical thrombectomy for distal occlusions: efficacy, functional and safety outcomes: insight from the STAR collaboration. World Neurosurg 2021;151:e871-e879.


31. Filioglo A, Simaan N, Honig A, Heldner MR, von Rennenberg R, Pezzini A, et al. Outcomes after reperfusion therapies in patients with ACA stroke: a multicenter cohort study from the EVATRISP collaboration. J Neurol Sci 2022;432:120081.


32. Dabhi N, Mastorakos P, Sokolowski J, Kellogg RT, Park MS. Mechanical thrombectomy for the treatment of anterior cerebral artery occlusion: a systematic review of the literature. AJNR Am J Neuroradiol 2022;43:1730-1735.


33. Meyer L, Stracke CP, Jungi N, Wallocha M, Broocks G, Sporns PB, et al. Thrombectomy for primary distal posterior cerebral artery occlusion stroke: the TOPMOST study. JAMA Neurol 2021;78:434-444.

34. Meyer L, Stracke P, Wallocha M, Broocks G, Sporns P, Piechowiak EI, et al. Aspiration versus stent retriever thrombectomy for distal, medium vessel occlusion stroke in the posterior circulation: a subanalysis of the TOPMOST study. Stroke 2022;53:2449-2457.


35. Saber H, Desai SM, Haussen D, Al-Bayati A, Majidi S, Mocco J, et al. Endovascular therapy vs medical management for patients with acute stroke with medium vessel occlusion in the anterior circulation. JAMA Netw Open 2022;5:e2238154.



36. Haussen DC, Eby B, Al-Bayati AR, Grossberg JA, Rodrigues GM, Frankel MR, et al. A comparative analysis of 3MAX aspiration versus 3 mm Trevo Retriever for distal occlusion thrombectomy in acute stroke. J Neurointerv Surg 2020;12:279-282.


37. Kühn AL, Wakhloo AK, Lozano JD, Massari F, De Macedo Rodrigues K, Marosfoi MG, et al. Two-year single-center experience with the ‘Baby Trevo’ stent retriever for mechanical thrombectomy in acute ischemic stroke. J Neurointerv Surg 2017;9:541-546.


38. Rikhtegar R, Mosimann PJ, Weber R, Wallocha M, Yamac E, Mirza-Aghazadeh-Attari M, et al. Effectiveness of very low profile thrombectomy device in primary distal medium vessel occlusion, as rescue therapy after incomplete proximal recanalization or following iatrogenic thromboembolic events. J Neurointerv Surg 2021;13:1067-1072.



39. Bourcier R, Mazighi M, Labreuche J, Fahed R, Blanc R, Gory B, et al. Susceptibility vessel sign in the ASTER trial: higher recanalization rate and more favourable clinical outcome after first line stent retriever compared to contact aspiration. J Stroke 2018;20:268-276.




40. Mohammaden MH, Haussen DC, Perry da Camara C, Pisani L, Olive Gadea M, Al-Bayati AR, et al. Hyperdense vessel sign as a potential guide for the choice of stent retriever versus contact aspiration as first-line thrombectomy strategy. J Neurointerv Surg 2021;13:599-604.


41. Kaesmacher J, Bellwald S, Dobrocky T, Meinel TR, Piechowiak EI, Goeldlin M, et al. Safety and efficacy of intra-arterial urokinase after failed, unsuccessful, or incomplete mechanical thrombectomy in anterior circulation large-vessel occlusion stroke. JAMA Neurol 2020;77:318-326.



42. Mattle HP, Arnold M, Georgiadis D, Baumann C, Nedeltchev K, Benninger D, et al. Comparison of intraarterial and intravenous thrombolysis for ischemic stroke with hyperdense middle cerebral artery sign. Stroke 2008;39:379-383.


43. Renú A, Millán M, San Román L, Blasco J, Martí-Fàbregas J, Terceño M, et al. Effect of intra-arterial alteplase vs placebo following successful thrombectomy on functional outcomes in patients with large vessel occlusion acute ischemic stroke: the CHOICE randomized clinical trial. JAMA 2022;327:826-835.

44. Cimflova P, Kappelhof M, Singh N, Kashani N, Ospel JM, McDonough R, et al. Factors influencing thrombectomy decision making for primary medium vessel occlusion stroke. J Neurointerv Surg 2022;14:350-355.