Introduction
Carotid artery stenosis or occlusion (CASO) is a major etiological factor for cerebral infarction that can cause cognitive deterioration, even without the development of symptomatic ischemic stroke. Additionally, CASO causes vascular cognitive impairment (VCI). Asymptomatic carotid artery stenosis of ≥50% can cause VCI even in the absence of focal symptoms and induce morphological change in the brain such as reducing cortical thickness [1-4]. Therefore, carotid artery stenosis of ≥50% increases the risk of VCI, indicating that asymptomatic CASO carries symptomatic implications. However, a large population exists with undetected cognitive morbidities. This finding has important public health implications. Moreover, the prevention or treatment of VCI, which may result from hypoperfusion in patients with CASO, is an urgent concern.
Resveratrol (3,5,4'-trihydroxy-trans-stilbene) is a polyphenol that is naturally occurring in the skin of red grapes, wine, blueberries, and peanuts [5]. Resveratrol plays an important role in the inhibition of various human diseases, such as brain disorders, via pleiotropic effects [6]. In the preclinical stage, resveratrol improved cognitive performance in rat models subjected to bilateral common carotid artery occlusion, mimicking chronic cerebral hypoperfusion by suppressing oxidative stress and inflammation and promoting autophagy [7-9]. Furthermore, the longevity gene SIRT1 which can be activated by resveratrol [10] revealed cognitive improvement and cerebral blood flow (CBF) preservation via deacetylation of endothelial nitric oxide synthesis (eNOS) in a mouse CASO model [11,12]. However, clinical evidence is limited, and only a few reports are available on the positive effects on cognition in healthy participants. Daily intake of oral resveratrol 200 mg/day for 26 weeks significantly improved memory and hippocampal functional connectivity in healthy participants aged 50-75 years [5]. Another study revealed that healthy postmenopausal women aged 45-85 years who received resveratrol 150 mg/day for 14 weeks displayed significant improvements in verbal memory and overall cognitive function [13]. However, the efficacy of resveratrol in asymptomatic patients with CASO remains unclear.
This study aimed to evaluate the association between long-term resveratrol intake and improvements in cognitive performance, with a focus on cerebral hemodynamic state preservation on 15O-gas positron emission tomography (15O-gas PET) in patients with asymptomatic CASO.
Methods
Study design
This retrospective cohort study was conducted at the National Cerebral and Cardiovascular Center (NCVC) of Japan, in accordance with the Declaration of Helsinki. The study was approved by the Research Ethics Committee of the NCVC (approval number: R20113). Patient participation was obtained through an optout methodology, which meant that participants were included in the study unless they expressed their decision to be excluded. The inclusion criteria were as follows: (1) patients with asymptomatic CASO who presented with a peak systolic velocity of >130 cm/s at a stenotic lesion or occlusion on carotid Doppler ultrasonography; (2) patients who underwent 15O-gas PET for the assessment of the CBF, cerebral metabolic rate of oxygen (CMRO2), and oxygen extraction fraction (OEF), and neuropsychological examinations including Montreal Cognitive Assessment (MoCA) and Alzheimer’s Disease Assessment Scale-Cognitive Subscale 13 (ADAS-Cog) twice between July 2020 and March 2022 with an interval of >125 days between visit 1 and visit 2 (Supplementary Figure 1). A peak systolic velocity of >130 cm/s at a stenotic lesion indicates a stenotic diameter of approximately ≥50%, which is comparable to moderate or severe stenosis [14,15]. Asymptomatic status was confirmed by the absence of stroke or transient ischemic attack attributed to the development of a carotid lesion within 180 days, with a modified Rankin Scale score ≤1 [16]. Patients with ≥60% asymptomatic carotid stenosis are at a high risk of stroke, even when receiving the best medical treatment [17]. Therefore, patients with asymptomatic moderate or severe CASO generally need to undergo functional brain imaging such as PET to validate whether surgical revascularization is indicated. Patients who received management strategies for metabolic and vascular risk factors and orally received 30 mg/day resveratrol (resveratrol group) and those who received management of metabolic and vascular risk factors without resveratrol (non-resveratrol group) at our neurology clinic were compared. The resveratrol group was administered the polyphenol at a dose of 30 mg/day (DHC Corporation, Tokyo, Japan) after visit 1 (Supplementary Figure 1). Data on medical history, blood test results, and medications were collected. The primary outcome was cognitive preservation or improvement following the resveratrol treatment. The secondary outcome was the preservation or improvement of cerebral hemodynamic values on 15O-gas PET.
15O-gas PET measurements
All patients underwent a series of 15O-gas PET examinations to assess CBF, CMRO2, and OEF (Supplementary Figure 2A). Radioactive 15O was produced by accelerating a deuteron (d) beam via a 14N(d,n)15O nuclear reaction using a cyclotron (CYPRIS HM-12, Sumitomo Heavy Industry, Tokyo, Japan). Furthermore, 0.3% oxygen (O2) in the nitrogen (N2) target gas was used to produce 15O-O2 and 15O-carbon monoxide (CO) gases, while 1.0% carbon dioxide (CO2) in the N2 target gas was used to produce 15O-CO2 gas.
A PET scanner (Biograph mCT; Siemens Healthineers, Erlangen, Germany) was used in this study. A 15O-gas PET scan was initiated 3 min after 2-min inhalation of 15O-CO inhalation for 4 min. An additional dynamic 15O-gas PET scan was performed for 8 min during sequential inhalation of 15O-O2 and 15O-CO2 gases for 1 min each at 4.5-min intervals. The radiochemical purity was confirmed to be >99% before radio gas inhalation in each patient via two-channel rapid gas chromatography (Micro 990, Agilent Technologies, Inc., Santa Clara, CA, USA), as described in a previous study [18].
15O-gas PET images were reconstructed using vendor software with an adequately selected methodology that considered the presence of gaseous 15O-radioactivity surrounding the face during the inhalation period [19]. Functional images of CBF, CMRO2, and OEF were obtained according to a previously validated technique (dual-table autoradiography technique) [20]. The arterial input function was obtained from the radioactivity concentration in the arterial blood, which was continuously withdrawn from the brachial artery [20]. The metabolized 15O-water in the arterial blood generated from 15O-O2 was estimated by modeling the physiological oxygen metabolism [21].
Neuropsychological assessment
All patients underwent neuropsychological examinations, including the MoCA and ADAS-Cog, performed by senior clinical neuropsychologists (CK and MY) blinded to the intervention groups.
Concentration of resveratrol and its metabolites
The concentrations of resveratrol and its metabolites were measured via liquid chromatography-mass spectrometry in patients who provided written informed consent to participate in the NCVC biobank. For a detailed description, see the Supplementary Methods.
Statistical analysis
All 15O-gas PET images were anatomically standardized using linear and nonlinear transformations with SPM2 [22] to ensure that the brain images of all patients had the same anatomical format. Three-dimensional (3D) Stereotaxic Region-of-interest (ROI) Template (3DSRT, PDRadiopharma Inc., Tokyo, Japan) was used (Supplementary Figure 2B) [23,24]. Twelve ROI segments grouped according to arterial supply were examined in each hemisphere: callosomarginal, pericallosal, precentral, central, parietal, angular, temporal, posterior, hippocampus, lenticular nucleus, thalamus, and cerebellum [25] (Supplementary Figure 2B). The entire hemisphere comprised the area, except for the ipsilateral cerebellum. The frontal lobe comprises the callosomarginal, precentral, and central artery areas. The counts under each ROI were normalized to the average count within the bilateral cerebellum to quantify relative CBF, CMRO2, and OEF.
Continuous variables with a normal distribution were expressed as means±standard deviations. Differential analysis was performed by subtracting each cognitive subscale and the total MoCA and ADAS-Cog scores, as well as the relative CBF, CMRO2, and OEF in each ROI between the time points to compare temporal changes in cognitive functions and cerebral hemodynamics between the two groups using the Student’s t-test. To identify independent predictors of cognitive function and CBF preservation in patients with CASO, multivariate linear regression analyses were conducted after adjusting for age, sex, hypertension, diabetes mellitus, dyslipidemia, and coronary artery disease, as appropriate. No data regarding the primary and secondary outcomes was missing. All reported P-values were two-tailed, and P-values <0.05 were considered statistically significant. All analyses were performed using the SPSS software version 27 (IBM Corp., Armonk, NY, USA) and GraphPad PRISM (Ver 8.4.3; GraphPad Software, Boston, MA, USA).
Results
Baseline characteristics of the participants
This study enrolled 79 asymptomatic patients with moderate or severe CASO who underwent neuropsychological assessments and 15O-gas PET. Among the 79 patients, 43 received the best medical treatment for metabolic and vascular risk factors and 36 received resveratrol 30 mg/day. The mean age (76.4±7.4 vs. 77.0±8.6 years) and the proportion of male patients (34 [79.1%] vs. 27 [75.0%]) were comparable between the non-resveratrol and resveratrol groups. The baseline mean total MoCA scores of the non-resveratrol and resveratrol groups were 23.6±3.6 and 23.7±3.6, respectively. Moreover, the baseline mean total ADAS-Cog scores were 14.6±5.4 and 15.8±7.2 in the non-resveratrol and resveratrol groups, respectively. Hence, these patients present with mild cognitive impairment (MCI) or mild dementia [26]. The mean observational periods between visit 1 and visit 2 in Supplementary Figure 1 were 244.8±86.9 and 221.2±61.9 days (P=0.18) (Table 1).
Resveratrol intake and blood concentration of resveratrol and its metabolites
In humans, oral resveratrol is rapidly metabolized into glucuronide and sulfate conjugates, with the parent molecules and metabolites initially peaking in the plasma 30-90 minutes after dosing [27,28]. The resveratrol group demonstrated a significant increase in blood concentrations of resveratrol and its metabolites, including resveratrol 3-O-glucuronide, resveratrol 4-O-glucuronide, and resveratrol 3-O-sulfate (Supplementary Table 1). The mean interval (±standard deviation) between the time of resveratrol intake and that of blood collection was 4 h 6 min (±3 h 49 min). Therefore, the blood concentration of resveratrol was substantially lower than that of its metabolites. We assumed that the resveratrol group consumed resveratrol daily as displayed in Supplementary Table 1.
Resveratrol intake and vascular risk factors
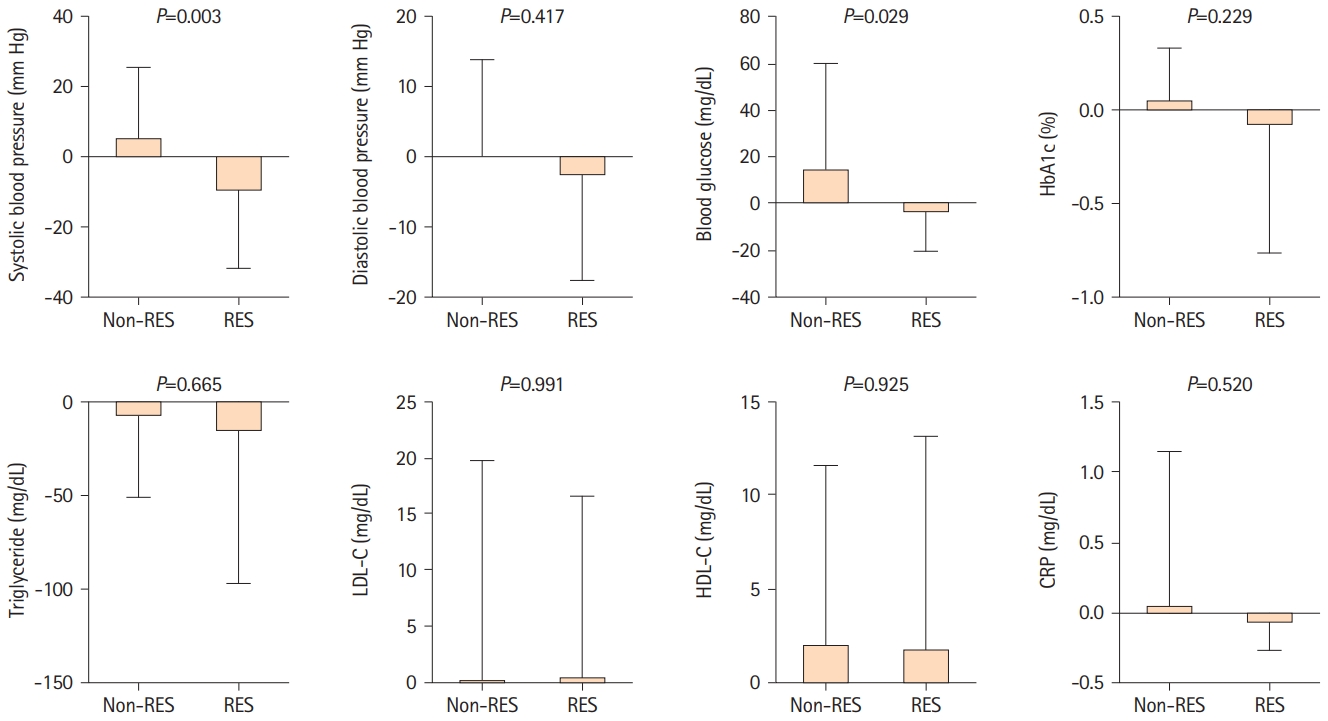
Figure 1 illustrates the longitudinal changes in blood pressure and blood test findings. The systolic blood pressure of the resveratrol group significantly decreased (-10.1±21.3 mm Hg), meanwhile, the systolic blood pressure of the non-resveratrol group increased slightly (5.8±19.8 mm Hg) (P=0.003). The routine blood glucose level of the resveratrol group significantly decreased by -3.0±16.9 mg/dL. However, the routine blood glucose level of the non-resveratrol group increased by 15.0±44.9 mg/dL (P=0.029). Diastolic blood pressure (P=0.417) and glycated hemoglobin (HbA1c) levels (P=0.229) in the resveratrol group were more likely to improve (Figure 1). No significant changes were observed in lipid metabolism, such as in triglyceride, low-density lipoprotein cholesterol, or high-density lipoprotein cholesterol levels (Figure 1).
Association between resveratrol intake and cognitive improvement in patients with asymptomatic CASO
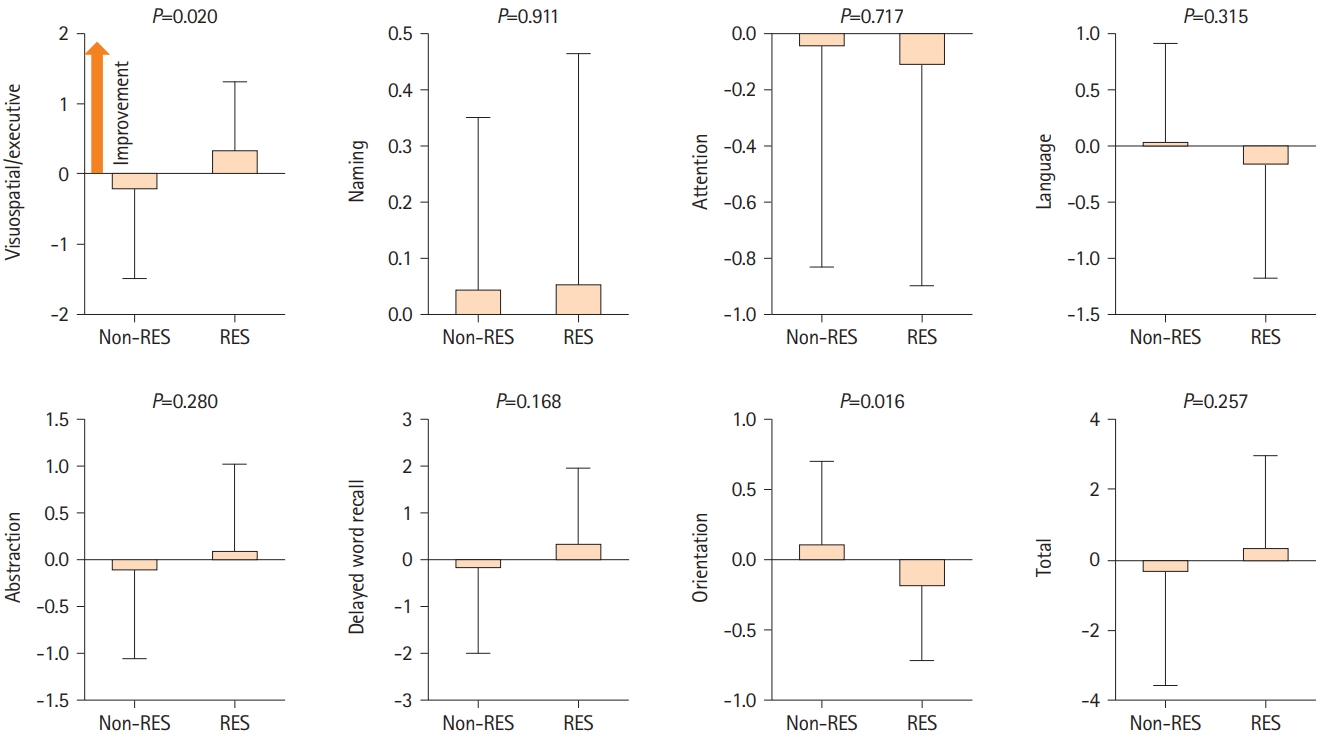
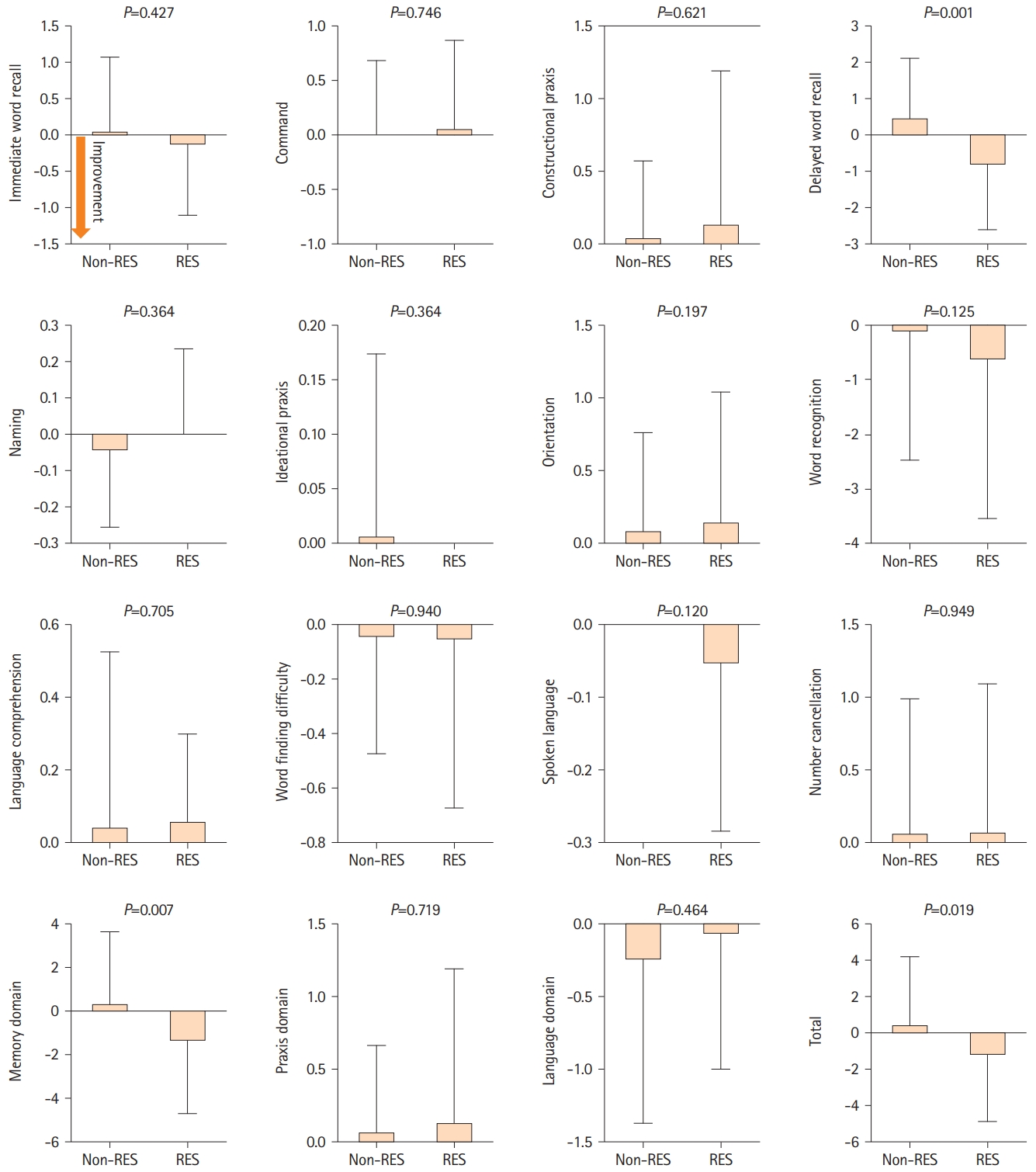
We examined the MoCA and ADAS-Cog scores to investigate cognitive changes related to resveratrol treatment. CASO mainly induces visuospatial, executive, and memory impairment [4,16]. Resveratrol intake was significantly associated with visuospatial/executive improvement (non-resveratrol group: -0.2±1.3 vs. resveratrol group: 0.4±0.9; P=0.020) based on MoCA. However, the MoCA total score did not demonstrate a significant difference (-0.4±3.2 vs. 0.4±2.6) (Figure 2). Furthermore, according to ADAS-Cog, a significantly high interval change was observed in the delayed word recall (0.5±1.6 vs. -0.9±1.8; P=0.001), memory domain (0.4±3.3 vs. -1.6±3.1; P=0.007), and total score (0.5±3.7 vs. -1.5±3.4; P=0.019) between the non-resveratrol and resveratrol groups (Figure 3). Therefore, multivariate linear regression model analysis was performed after adjusting for age, sex, hypertension, diabetes, dyslipidemia, and coronary artery disease. Resveratrol intake was an independent predictor of improvement in visuospatial/executive function (β=0.26, 95% confidence interval [CI]: 0.06-1.15, P=0.030) and memory (memory domain: β=-0.26, 95% CI: -3.23 to -0.15, P=0.032; delayed word recall: β=-0.37, 95% CI: -2.16 to -0.54, P=0.001) (Table 2). Thus, resveratrol may contribute to the amelioration of visuospatial, executive, and memory impairments in patients with CASO.
Association between resveratrol intake and CBF improvement in patients with asymptomatic CASO
To identify the reason for the significantly high interval change in visuospatial and executive functions and memory in the resveratrol group, cerebral hemodynamics, including relative CBF, CMRO2, and OEF, were assessed using 15O-gas PET. Initially, the CBF changes in each hemisphere were analyzed. Compared with the non-resveratrol group, the resveratrol group was more likely to present with CBF improvement in the entire right (0.0011±0.034 vs. 0.016±0.032; P=0.051) and left (-0.0011±0.035 vs. 0.014±0.031; P=0.054) hemispheres. To examine the aforemention changes in detail, we analyzed CBF in the frontal lobe, basal ganglia, and thalamus, which govern visuospatial and executive function [29] and memory. Longitudinally, long-term resveratrol intake was associated with a significant increase in relative CBF in the right frontal lobe (non-resveratrol group: 0.001±0.037 and resveratrol group: 0.019±0.033; P=0.027) (Figure 4). In the frontal lobe, the right (-0.004±0.041 vs. 0.018±0.034; P=0.013) and left (-0.003±0.040 vs. 0.017±0.042; P=0.031) callosomarginal artery areas, and the right precentral artery area (0.002±0.040 vs. 0.021±0.035; P=0.028) demonstrated associations between significant increased CBF and long-term resveratrol intake (Figure 4). Furthermore, the left lenticular nucleus (-0.002±0.06 vs. 0.033±0.06; P=0.009), right thalamus (-0.002±0.085 vs. 0.039±0.85; P=0.035), and left thalamus (-0.011±0.095 vs. 0.045±0.090; P=0.010) also demonstrated significant associations (Figure 4). After adjusting for age, sex, hypertension, diabetes, dyslipidemia, and coronary artery disease, the multivariate linear regression models displayed that long-term resveratrol intake was an independent predictive factor of increased CBF in the right frontal lobe (β=0.25, 95% CI: 0.001-0.035; P=0.039), the right callosomarginal artery area (β=0.29, 95% CI: 0.005-0.041; P=0.013), the right precentral artery area (β=0.25, 95% CI: 0.001-0.037; P=0.042), the left lenticular nucleus (β=0.31, 95% CI: 0.009-0.067; P=0.011), and the left thalamus (β=0.29, 95% CI: 0.011-0.102; P=0.016) in patients with asymptomatic CASO (Table 3). The relative CMRO2 changes were comparable between the two groups in the entire right (0.0044±0.035 vs. 0.0095±0.037; P=0.53) and left (0.0033±0.035 vs. 0.0069±0.033; P=0.65) hemispheres as well as in the bilateral frontal lobe, lenticular nucleus, and thalamus (Supplementary Figure 3). Similarly, the relative OEF changes were also comparable between the two groups in the entire right (-0.0028±0.39 vs. -0.0040±0.48; P=0.91) and left (-0.0028±0.42 vs. -0.0001±0.48; P=0.79) hemispheres as well as in the bilateral frontal lobe, lenticular nucleus, and thalamus (Supplementary Figure 4). Thus, long-term resveratrol intake may contribute not only to cognitive improvement but also to increased CBF. No adverse events were reported.
Discussion
This study demonstrated that long-term intake of oral resveratrol might improve cognitive functions, including visuospatial and executive function, and memory. Furthermore, CBF was associated with cognitive function in asymptomatic patients with moderate or severe CASO.
Currently, no promising preventive or treatment strategies are available for CASO-related VCI. Carotid revascularization, such as carotid endarterectomy (CEA) and artery stenting (CAS), does not affect cognition, although restores cerebral perfusion. However, operative ischemia, including subclinical cerebral microembolic patterns that occur during revascularization, may worsen neuropsychological function [30]. One meta-analysis [31] revealed that CEA and CAS did not improve cognitive dysfunction. To prevent cognitive deterioration in patients with CASO, cerebrovascular integrity should be maintained without perioperative cerebral ischemia or silent microembolic cerebral injury. Furthermore, no evidence supports the efficacy of antithrombotic drugs in preventing VCI [32].
In this setting, where no novel candidates had been established, the first report demonstrating that resveratrol treatment preclinically improved cerebrovascular dysfunction was published in 2014. Resveratrol preserves neurovascular function by reducing oxidative stress and improving vascular endothelial function in 24-month-old mice [33]. Thereafter, several in vivo studies have displayed that resveratrol improves cognitive performance via pleiotropic effects, including suppression of oxidative stress and inflammation, promotion of autophagy, and anti-apoptotic effects in rat models subjected to bilateral common carotid artery occlusion mimicking chronic cerebral hypoperfusion. For example, resveratrol activates autophagy and inhibits the expression of protein kinase B (AKT)/mechanistic target of rapamycin signaling pathway-related proteins, thereby reducing oxidative stress-induced neuronal damage in the frontal cortex and hippocampus and neuronal apoptosis [7]. Resveratrol can improve cognitive function by activating protein kinase A and cyclic adenosine monophosphate (cAMP) responsive element-binding protein pathways, which prevent the inhibition of long-term potentiation and restore dendritic spinal impairment in rats [8]. Resveratrol reduces the inflammatory response during chronic cerebral hypoperfusion by downregulating the stimulator of interferon genes/TRAF family member-associated NF-κB activator (TANK)-binding kinase 1/interferon regulatory factor 3 signaling [9].
Alternatively, resveratrol may activate the nicotinamide adenine dinucleotide (NAD+)/Sirtuin 1 (SIRT1) pathway [10]. Resveratrol competitively inhibits cAMP-degrading phosphodiesterases, leading to elevated cAMP levels. Therefore, resveratrol increases NAD+, resulting in increased SIRT1 activity [10]. Resveratrol also activates eNOS via SIRT1 activation [34]. Our previous study demonstrated that working memory, CBF, and white matter integrity were preserved in Sirt1-transgenic mice with bilateral common carotid artery stenosis. Brain eNOS was deacetylated in Sirt1-transgenic mice, leading to CBF and cognitive restoration [11]. In addition, we provided evidence for the promising protective role of SIRT1 against bilateral common carotid artery occlusion by preserving CBF and restoring the cerebrovascular reserve in mice [12]. Thus, resveratrol may contribute to the aforementioned beneficial effects in the cerebrovascular system by activating the NAD+/ SIRT1-eNOS axis, which is the main pathway.
Notably, the resveratrol group demonstrated a significant reduction in systolic blood pressure. Blood pressure can decrease via endothelial restoration associated with the activation of the NAD+/SIRT1-eNOS axis. Oral resveratrol supplementation improved endothelium-dependent vasodilation, as demonstrated by a significant increase (approximately 1.5 times) in flow-mediated dilation at a dose of 30 mg/day relative to the placebo [35]. Therefore, resveratrol was administered at a dose of 30 mg/day in this study. Therefore, resveratrol may be effective in treating patients with hypertension. Furthermore, resveratrol intake was associated with decreased blood glucose and HbA1c levels in this study. Resveratrol potentially improves glucose delivery, uptake, and substrate utilization for energy production and reduces circulating blood glucose and insulin levels via eNOS activation and glucose transporter-4 translocation [36].
This study demonstrated that cognitive improvement in the resveratrol group accompanied an increase of approximately 1.5%-7% in relative CBF (Supplementary Table 2). Methylthionine (138 mg/day), a tau aggregation inhibitor, induced an increase of approximately 1.5%-2.5% in CBF normalized to the cerebellum throughout the entire brain and a significantly larger ADAS-Cog effect size than the placebo in patients with mild Alzheimer’s disease [37]. Therefore, such increase in the CBF may be clinically beneficial for cognitive improvement.
In this study, CBF, CMRO2, and OEF values were normalized to the cerebellum to minimize the influence of inter-examination variability on repeated testing. This adjustment accounts for potential differences in whole-brain counts that may arise due to variations in the depth of breathing during different 15O-gas PET examinations [38]. Studies have demonstrated that relative CBF normalized to the cerebellum is employed in patients with Alzheimer’s disease and Moyamoya disease [37,39,40], which are not expected to affect cerebellar perfusion. Moreover, 3D stereotactic surface projection, which enables quantitative data extraction and reliable localization of abnormalities through stereotaxic coordinates using the cerebellum as a reference region, has been widely used to improve the diagnostic performance of PET [41]. Thus, cerebellar normalization is a beneficial method for minimizing the influence of inter-examination variability during repeated testing using brain 15O-gas PET.
This study has several limitations. First, patients receiving the placebo control were not compared with those receiving regular health advice and medications for controlling vascular risk factors. Based on these results, randomized, double-blind, placebo-controlled trials should be performed in the near future. Second, the sample size was small; thus, randomized controlled trials with large sample sizes must be conducted. Despite these limitations, our results revealed that cognitive function and CBF may be maintained or improved with long-term oral resveratrol intake in asymptomatic patients with moderate or severe CASO who present with cognitive functions ranging from MCI to mild dementia.
Conclusions
In conclusion, cognitive function and CBF could be maintained or improved with long-term intake of oral resveratrol, which has pleiotropic neuroprotective effects such as eNOS activation, in patients with asymptomatic moderate or severe CASO who have cognitive function ranging from MCI to mild dementia. As no disease-modifying therapies for dementia are available, including VCI, preventive therapies must be developed. Our results emphasize the need for a randomized, double-blind, placebo-controlled trial to analyze the effects of resveratrol in preserving cognitive function in patients with VCI caused by asymptomatic moderate or severe CASO.