Clot Composition Analysis as a Diagnostic Tool to Gain Insight into Ischemic Stroke Etiology: A Systematic Review
Article information
Abstract
Mechanical thrombectomy renders the occluding clot available for analysis. Insights into thrombus composition could help establish the stroke cause. We aimed to investigate the value of clot composition analysis as a complementary diagnostic tool in determining the etiology of large vessel occlusion (LVO) ischemic strokes (International Prospective Register of Systematic Reviews [PROSPERO] registration # CRD42020199436). Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, we ran searches on Medline (using the PubMed interface) and Web of Science for studies reporting analyses of thrombi retrieved from LVO stroke patients subjected to mechanical thrombectomy (January 1, 2006 to September 21, 2020). The PubMed search was updated weekly up to February 22, 2021. Reference lists of included studies and relevant reviews were hand-searched. From 1,714 identified studies, 134 eligible studies (97 cohort studies, 31 case reports, and six case series) were included in the qualitative synthesis. Physical, histopathological, biological, and microbiological analyses provided information about the gross appearance, mechanical properties, structure, and composition of the thrombi. There were non-unanimous associations of thrombus size, structure, and composition (mainly proportions of fibrin and blood formed elements) with the Trial of Org 10172 in Acute Stroke Treatment (TOAST) etiology and underlying pathologies, and similarities between cryptogenic thrombi and those of known TOAST etiology. Individual thrombus analysis contributed to the diagnosis, mainly in atypical cases. Although cohort studies report an abundance of quantitative rates of main thrombus components, a definite clot signature for accurate diagnosis of stroke etiology is still lacking. Nevertheless, the qualitative examination of the embolus remains an invaluable tool for diagnosing individual cases, particularly regarding atypical stroke causes.
Introduction
Mechanical thrombectomy has not only become the standard of care in the management of most large vessel occlusion (LVO) strokes, but it also renders the occluding clot available for lab bench analysis [1]. Insights into thrombus composition and properties could help determine its relationships with the clot signs on imaging, stroke cause, resistance to thrombectomy, procedural complications, and outcome measures [2].
A previous systematic review of studies published between January 2005 and December 2015 on imaging and histologic characteristics of thrombi in acute ischemic stroke (AIS) concluded that the hyperdense artery sign was associated with red blood cell (RBC)-rich thrombi and improved recanalization rates. However, there was no association between the histopathological characteristics of thrombi, stroke etiology, and angiographic outcomes [3]. Another recent scoping systematic review focused on the impact of thrombus composition on the efficacy of mechanical thrombectomy and thrombolysis [4]. However, assessing the value of clot analysis in the diagnosis of stroke etiology and thus guiding secondary prevention strategies seems more challenging.
This systematic review aimed to evaluate the value of clot composition analysis as a complementary diagnostic tool in determining the LVO ischemic stroke etiology. Specifically, we addressed the following research questions: (1) What types of physical, histological, or other biological analyses have been carried out on thrombi retrieved from LVO stroke patients subjected to mechanical thrombectomy?; (2) What kind of information about the structure, and molecular and cellular composition of stroke thrombi has resulted from laboratory analyses?; (3) Could laboratory analyses of clot structure and composition be used as complementary diagnostic tools to determine stroke etiology, and thus reduce the proportion of cryptogenic strokes?; (4) Could specific findings in clot composition be used as ancillary information to diagnose atypical stroke etiologies due to underlying pathologies?
Methods
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Systematic review
This systematic review was carried out according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [5]. The protocol was registered in the CRD-NIHR International Prospective Register of Systematic Reviews (PROSPERO) with registration number CRD42020199436 [6].
We searched the published literature reporting the analysis of thrombi retrieved from LVO stroke patients subjected to mechanical thrombectomy and performed a qualitative assessment of the available evidence.
Literature search strategy
We carried out electronic searches on Medline (using the PubMed interface) and Web of Science from January 1, 2006, up to and including September 21, 2020. The search syntax was (stroke AND (thrombus OR thrombi OR clot)) AND (thrombectomy OR endovascular). The search fields were [Title/Abstract] in Medline (PubMed) and [Topic] in Web of Science. There were no language restrictions. The PubMed search was updated weekly through My NCBI up to February 22, 2021. References were added to a Mendeley Reference Manager library dedicated to this review’s topic, checked for duplicates, and completed with Mendeley’s feedback-delivering personalized suggestions for related articles. Reference lists of included studies and relevant reviews were hand-searched. The electronic database search was supplemented by searching for trial protocols through ClinicalTrials.gov Advanced Search syntax: condition or disease (ischemic stroke) and other terms ((thrombus OR thrombi OR clot) AND (thrombectomy OR composition)). The search was not extended to unpublished studies or other sources of grey literature.
Study selection: eligibility criteria and screening process
The current review considered observational cohort studies, case series and case report studies reporting any kind of physical, histological, or other biological analyses carried out on thrombi retrieved from LVO stroke patients subjected to mechanical thrombectomy. We included studies published as full-length original research articles in any language, provided that the English abstract was available, and abstracts of conference proceedings in English language. In cases of studies with duplicate or overlapping patient populations, only the publication with the most complete dataset was included. Protocol articles, review articles and abstracts later published in full were also excluded.
Titles and abstracts yielded by the search were independently screened against the inclusion criteria by two reviewers. Full reports were obtained for all titles that appeared to meet the inclusion criteria or where there was any uncertainty. Reviewer pairs then screened the full-text reports of potentially eligible studies and decided whether these met the inclusion criteria. Disagreements were resolved through discussion and consensus involving a third reviewer.
Data extraction and synthesis
The following information was collected from the eligible studies and extracted to tables independently by two reviewers: general information (first author name, year of publication, source, and type of study), type(s) of physical, histological, or other biological analyses carried out on thrombi, sample size(s), qualitative and quantitative features about thrombus composition, and diagnostic information regarding typical LVO ischemic stroke Trial of Org 10172 in Acute Stroke Treatment (TOAST) etiologies or atypical etiologies with underlying pathologies.
Heterogeneity in study design, outcome measuring, and reporting precluded a meta-analysis of the association between thrombus composition and typical or atypical stroke etiology. Instead, a systematic narrative synthesis is provided with information presented in the text and tables to summarize and explain the characteristics and findings of the included studies.
Results
Search results: study selection and characterization
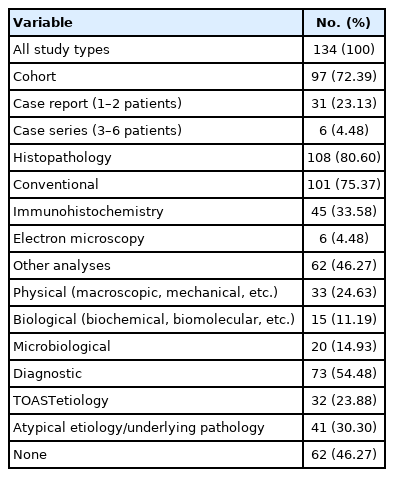
A detailed study selection flow chart is shown in Figure 1. Briefly, a total of 2,665 records were identified electronically in the Web of Science and Medline (through PubMed) databases up to and including September 21, 2020. After removing duplicate and irrelevant records, and adding relevant records identified through an updated search on PubMed (up to and including February 22, 2021) and other sources, 152 full-text articles and congress abstracts were assessed for eligibility. Subsequent reasoned exclusions rendered 134 studies which were included in the qualitative synthesis (Supplementary Table 1).
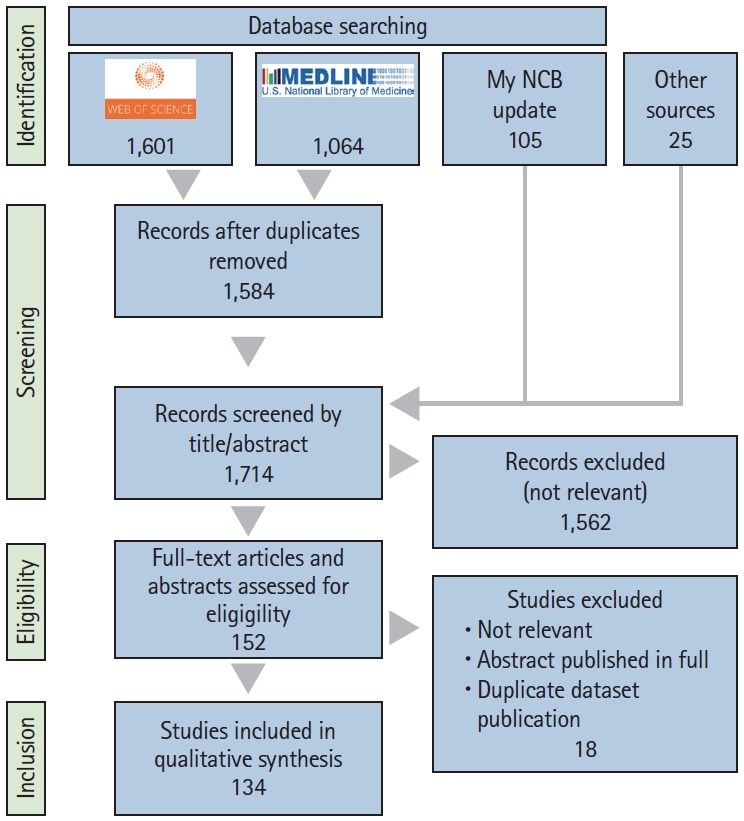
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart of the literature search results and selection of studies at each stage.
A summary of the study characteristics is shown in Table 1. Most of them (72.39%) were observational cohort studies with a wide range of cohort sizes, varying from four to 1,022 patients with thrombus samples subjected to analysis (median, 65 [interquartile range, IQR, 37 to 105]). Thirty-one (23.13%) case reports and six (4.48%) case series were also included. In 108 out of the 134 studies, the retrieved thrombi were subjected to one or more types of histopathological examinations. Physical, biological, and/or microbiological analyses were carried out in 62 studies. A relationship between thrombus structure/composition and TOAST or atypical etiologies was reported in 54.48% of the studies. In 46.27% of the studies, the stroke etiology was not taken into consideration, or non-conclusive results were obtained.
The publication chronology of the studies is shown in Figure 2. Although Marder’s pioneering study was published 15 years ago [7], 75.37% of the studies have been published in the last 5 complete years (2016 to 2020). Of note, 80% of the studies assessing thrombus composition by means of biological analyses using biochemical or molecular biology techniques have been published in the last 3 complete years (2018 to 2020). Since 11 studies have been published in 2021 up to and including February 22, around 75 studies are estimated to be published in the present year.
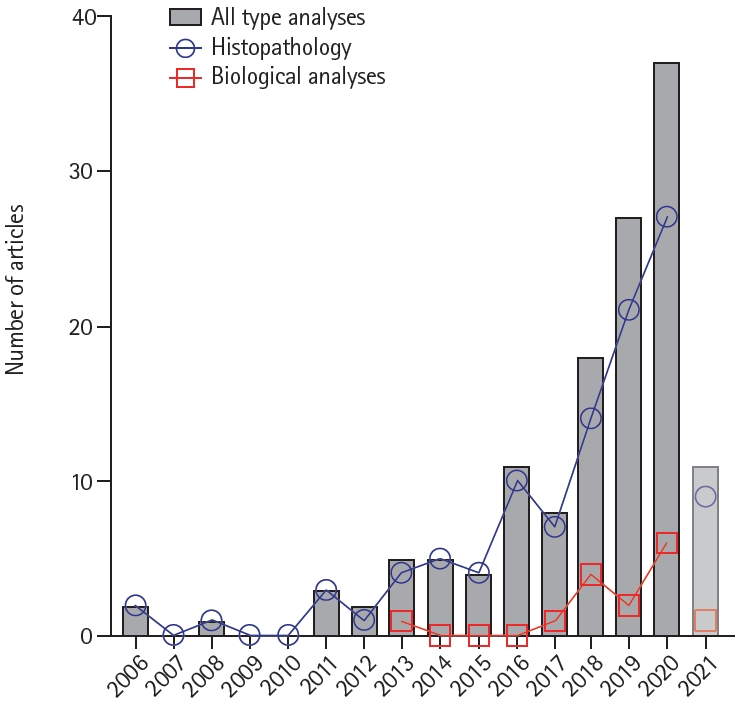
Chronology of study publication. Number of yearly-published studies from January 1, 2006, to February 22, 2021. Faded colors indicate incomplete year 2021.
The search for clinical trial protocols identified five ongoing studies in which thrombi and blood samples are being collected for histopathological and/or other biological analyses [8-12]. Concurrently, active multi-institutional registries like RESTORE (National University of Ireland) and Stroke Thromboembolism Registry of Imaging and Pathology, Mayo Clinic (STRIP) are compiling clinical, procedural, imaging, and histopathological data from patients with AIS.
Types of physical, histological, and other biological analyses
Procedures for thrombus retrieval and subsequent analysis have been reviewed [1,13]. Briefly, after retracting the thrombectomy device, the retrieved clot material is gently removed from the device and transferred into saline solution. If clot per-pass analysis is desired, clot material from each pass can be processed separately. The macroscopic appearance and other physical properties of the retrieved thrombus can be freshly examined; otherwise, the clot can be flash-frozen for storage and later biological analyses. For histological analysis, the specimens are fixed, paraffin-embedded, sectioned, and stained depending on the component of interest. Both manual quantifications and color-based segmentation analysis of thrombus components are used.
Supplementary Table 1 summarizes whether physical, histological, or biological analyses were carried out in each of the 134 included studies [7,14-146].
In 33 studies, thrombi were subjected to some kind of physical analysis. The gross appearance of the retrieved thrombus was reported in 29 studies. In case reports (20 studies) and case series (two studies), the macroscopic aspect of the thrombus was shown mostly through photographs. Mechanical behavior of the thrombi was analyzed in two studies using custom-made platforms and marketed devices, respectively. In one study the thrombi were scanned using high spatial-resolution three-dimensional (3D) T1-weighted magnetic resonance imaging (MRI) to reveal morphological and other physical characteristics. Finally, advanced synchrotron-based imaging techniques were used in one study to map thrombus composition.
Different types of histopathological examinations, including conventional histology, immunohistochemistry, and electron microscopy, were carried out in 108 studies. The most frequently used conventional stains were hematoxylin and eosin (H&E, 86 studies), Martius Scarlet Blue (MSB, 18 studies), Elastica van Gieson (14 studies), Prussian blue (eight studies), Masson’s trichrome (five studies), and Von Kossa (four studies). Other seldom-used stains were periodic-acid Schiff, Carstairs’ staining, Picro-Mallory, Alcian blue, Luna, Mallory’s phosphotungstic acid-hematoxylin, Ladewig trichrome, naphthol AS-D chloroacetate, and Feulgen’s reaction. Immunohistochemistry was carried out in 45 studies with specific antibodies against cellular and biomolecular thrombus components. Finally, the thrombi composition and organization were analyzed at the ultrastructural level through electron microscopy in six studies.
Regarding biological analyses, thrombus composition was assessed through different biochemical, biomolecular, and cellular techniques, such as enzymatic assay, enzyme-linked immunosorbent assay (ELISA), transcriptomics (real-time reverse transcription polymerase chain reaction [qRT-PCR]), proteomics, metabolomics, and flow cytometry in 15 studies. Finally, microbiological analysis of the thrombi was performed through classical culture techniques, specific staining for bacteria/fungi and/or metagenomics (qPCR) in 20 studies.
Thrombus gross appearance and mechanical properties
Case reports and case series, usually presenting thrombectomies of LVOs of atypical etiologies, showed mostly photographic images of the thrombus, and described whether single or multiple clots were retrieved, as well as their gross appearance, including size, shape, consistency, visual texture, color, and homo/heterogeneous aspect [14-34].
Macroscopic analysis of retrieved thrombi was also carried out in some cohort studies and one case series. The clot color was categorized using three different terminologies. Two cohort studies classified the thrombus as “red-black” or “white,” showing similar results: 94.2% red-black thrombi versus 5.8% white thrombi [35]; and 91.4% red/black thrombi versus 8.6% white thrombi [36]. Another cohort study categorized the thrombi by visual assessment as pinkish (17.3%), red (53.8%), or dark red (28.8%) [37]. A fourth cohort study described the thrombi as slightly white, darker, or reddish, but did not report percentages [38]. Regarding thrombus size, other cohort studies focused on the total extracted clot area (ECA) for each case, defined as the sum of the clot areas from all clot fragments within a case [39]. In a cohort of 550 patients, the mean ECA for all cases was 64 mm2 and the median number of fragments per case was 3 [40]. Of note, bridging therapy was associated with the retrieval of significantly smaller clots [41,42]. Median ECA was 0.33 cm2 (IQR, 0.16 to 0.59) in patients pre-treated with alteplase (recombinant tissue plasminogen activator [rtPA]), versus 0.39 cm2 (IQR, 0.22 to 0.82) in patients treated with mechanical thrombectomy alone [41]. Finally, in a case series of five patients the median volume of the thrombus, calculated using the ellipsoid formula (4/3πr1r2r3), was 10.47 mm3 [43].
Only two studies with limited sample sizes evaluated the mechanical behavior of the thrombi. The specimens were mechanically heterogeneous, in line with the histological heterogeneity. Stiffness and elasticity were measured with a dynamic mechanical analyzer, showing that red thromboemboli composed mainly of fibrin (FBR) and RBC were much softer than the calcified and cholesterol-rich material [44]. The tensile strength and response to stress were measured with a quasistatic uniaxial tensile test using a custom-made platform. The ultimate tensile strain of the emboli increased with a higher platelet (PLT) percentage, and the ultimate tensile stress increased with a higher FBR percentage and decreased with a higher RBC percentage [45].
Thrombus structure and composition: advanced imaging and histopathology
Multiparametric MRI has been used to characterize retrieved cerebral thrombi. Preliminary results showed that T1-weighted images with the corresponding apparent diffusion coefficient (water mobility) and T2 maps (relaxation time) could be used to assess thrombus compactness and microstructure, which in turn reflect RBC and PLT/FBR meshwork content [46]. Advanced synchrotron-based imaging techniques, including X-ray fluorescence and Fourier-transform infrared spectroscopy, have been used in freshly retrieved thrombi to map the distribution of biological elements and metabolites, respectively [47].
Most histopathology studies focused on the presence and relative abundance of FBR, and blood formed elements (RBCs, PLTs, and white blood cells [WBCs]) in the thrombus. Classical H&E staining was almost always used to visualize the general structure of the thrombus. Although H&E staining and machine learning software allowed reproducible quantification of the three major clot components (RBCs, WBCs, and FBR) [48], the more specific MSB staining was used in many studies for the selective quantification of RBCs and FBR [28,37,39,43,44,49-61]. The Picro-Mallory stain was used to assess the maturity of FBR in thrombi, based on its age-dependent differential staining [62]. Immunohistochemical staining procedures allowed the best visualization and reliable separate quantification of RBCs (glycophorin A, also designated CD235a antigen) [63-65], WBCs (CD45 antigen) [66-70], PLTs (CD41, CD42b, and CD61 antigens) [38,45,49,51,53,56,57,60,63,65,66,71-73], and fibrinogen/FBR [49,53,63,65,66,74]. Depending on the discriminatory capability of the staining procedures used, the studies expressed the ratios of clot components according to four different classifications, as shown in Table 2. A number of studies categorized the clots according to the dominant component (usually with a 60% cut-off) [48] as RBCrich, FBR-rich, PLT-rich, or mixed [7,50,52,54,58,59,64,75-91]. Some studies identified and/or quantified WBC types and subtypes by immunohistochemical staining using specific antibodies against neutrophil elastase (NE) [66,76,92-94], neutrophil myeloperoxidase (MPO) [65,66,76,95], Ly6G (monocyte, granulocyte, and neutrophil) [94], CD3 (T lymphocyte) [66,68-70,90,96], CD4 (T lymphocyte) [52,53], CD14 (monocyte) [66], CD15 (neutrophil, eosinophil, and monocyte) [97], CD20 (B lymphocyte) [66,68-70,96], CD66b (neutrophil) [76,93], and CD68 (monocyte and macrophage) [30,52,67-70,96,98].
Neutrophils were the predominant leukocyte subset in stroke thrombi [66,93]. Neutrophil extracellular traps (NETs) have been identified as part of the clot scaffold by using antibodies against NETosis biomarkers (citrullinated histones) and histochemical staining of extracellular DNA [63,65,66,76,93-95,99]. NETs were visualized in almost all (79.1% to 100%) of the analyzed thrombi in different studies [63,65,66,76,93,95], in amounts ranging from 0.21% to 13.45% of total thrombus area [93], and 1.1%±2.7% on average [76]. NET signals were observed as being confined within cells, filopodia-like structures, or web-like structures [65], especially in the outer thrombus layers [63,95], and almost exclusively within FBR-rich areas [76]. Inside the NETs, citrullinated histones were co-localized with inflammasome proteins (caspase-1 and apoptosis-associated speck-like protein containing a caspase-recruitment domain [ASC]) [99], granular neutrophil proteins (MPO) and extracellular DNA released from neutrophils [93,95]. The addition of histone-DNA complexes to FBR resulted in thicker fibers accompanied by increased rigidity, which contributed to the structural complexity and stabilization of the thrombi [76]. Of interest, monocytes could also form extracellular traps, but to a lesser extent than neutrophils [66,94].
Coagulation system proteins other than fibrinogen/FBR were immunohistochemically identified by using specific antibodies against von Willebrand factor (VWF) [52-56,60,63,74,97,100], and two PLT-derived direct inhibitors of tissue plasminogen activator (tPA): plasminogen activator inhibitor-1 [63,74] and protease nexin-1 [63]. The proportion of VWF varied from 0.1% to 94.3% of the total clot area [60], with mean values between 11.8% and 29.8% in different studies [56,60,100], and higher content in thrombi retrieved after unsuccessful intravenous thrombolysis [74]. VWF levels were correlated with those of FBR and PLTs [97]. White FBR-rich thrombi showed higher percentages of VWF+ areas co-localized with regions of FBR/collagen [52]. Similarly, PLT-rich areas were characterized by dense FBR structures aligned with VWF [53].
The presence of other thrombus components has been assessed using specific staining procedures. Some cohort studies used standard protocols including H&E and one or more of the following staining procedures to visualize elastic collagen fibers (Elastica van Gieson), hemosiderin/iron (Prussian blue), calcifications (Von Kossa), and collagen (Masson’s trichro me) [67-70,78,81,86,88,89,91,96,101]. However, positive results showing the occasional presence of intimal collagen fibers, cholesterol clefts, and smooth muscle cells (immunohistochemically stained for α-smooth muscle actin) were reported only in three studies assessing thrombectomy-induced wall damage, which identified vascular wall components or atheromatous gruel in a low proportion (2.6% to 20%) of the retrieved thrombi [78,91,101]. Case reports also used these specific stainings to identify elastic fibers, hemosiderin granules, collagen fibrous stalks, calcium deposits, and myofibroblast-like cells, usually in atypical thrombi [15,23,30,43,102].
The presence of foreign bodies in thrombi has also been evaluated. Delaminated polymer coating particulates were readily detected in 33% of H&E-stained preparations [103]. Thread or sheet-like structures were found in 25% of thrombi scanned by electron microscopy [104].
Classical descriptions of thrombus histological patterns based on light microscopy categorized their organized structure as layered (PLT bands arranged in layers), serpentine (PLT bands arranged in a serpentine way), or erythrocytic (with RBCs and nucleated cells interspersed) [7,52,75,105,106]. The molecular and cellular organization revealed distinct features between clots as well as among different regions within a clot. Thrombi were composed of two main area types: RBC-rich and PLT-rich areas. RBC-rich areas had limited complexity and consisted of RBCs entangled in a meshwork of thin FBR. Conversely, PLT-rich areas were characterized by dense FBR structures aligned with VWF and abundant amounts of WBCs and DNA that accumulate around [53]. Similarly, serial block-face scanning electron microscopy (SEM) showed a thrombus 3D ultrastructure that varied greatly depending on the region analyzed. RBC-rich areas were composed mainly of tightly packed RBCs deformed into polyhedrocytes with scant FBR fibers interwoven between cells. The regions with mixed composition showed thick FBR fibers along with PLTs, WBCs, and RBC clusters. FBR-rich areas contained dense FBR masses with sparse RBCs [56]. High-resolution SEM and transmission electron microscopy revealed a dense, sealed, rtPA-resistant external shell encapsulating a loose RBC-rich core. Shell components were densely compacted and agglomerated and formed a continuous layer, in which individual cells could hardly be detected. This was in stark contrast to the clearly identifiable RBCs, FBR fibers, and aggregated PLTs in the inner core [63,107]. Other ultrastructural studies showed morphologic features consistent with the presence of NETs, calcified deposits and cholesterol crystals in the clots [44,99].
Microbial pathogens in thrombi
Regarding septic cerebral emboli, Marder’s pioneering study reported one case of mycotic embolus [7]. However, both real-time and standard PCR demonstrated no expression of bacterial 16S rDNA in any of the 20 clot samples. Gram staining results also showed no evidence of bacteria [108]. Contrastingly, bacteria were detected in Gram-stained clots of four out of 65 patients (6.2%) [109]. A larger cohort of 75 patients showed DNA signatures of oral streptococcal bacteria in 84% of the retrieved thombi [110]. Metagenomics analysis also showed the presence of bacterial DNA in all four thrombi originated from symptomatic carotid plaques [111].
Thrombus composition: molecular biology, biochemical assays, and flow cytometry
Clot homogenates subjected to qRT-PCR showed the expression of inflammatory cytokines (interleukin 1β [IL-1β], IL-6, IL-8, IL18, tumor necrosis factor α [TNF-α], and monocyte chemoattractant protein-1 [MCP-1]), matrix metalloproteinases-2, -9 [112], and endothelial CD31 [75]. Four-plex assay showed the expression of cytokines (IL-1β and IL-18) and other inflammasome signaling proteins such as caspase-1 and ASC. Moreover, the presence of nucleotide-binding oligomerization domain (NOD)-like receptor protein-1 and absent in melanoma-2, two receptors that interact with caspase-1 and ASC to form an inflammasome complex, was shown by immunoblot analysis [99].
Proteomic analysis has been used to characterize the protein cargo of thrombi (thousands) and commonly present proteins (hundreds). Functional bioinformatics analyses revealed protein clusters related to inflammation [113]; immunological functions, blood cell dependent functions, and peripheral vascular processes [114]; metabolic processes, inflammatory track, and cell proliferation, activation, or motility [115]; primary hemostasis, integrin and kinase signaling linked to integrins, glycolysis, and acute phase reactants [116]. Nevertheless, metabolomics analysis suggested clot sorbitol content as a surrogate marker reflecting blood glucose level at stroke onset [117].
PLT, RBC, and WBC content of AIS thrombi could be estimated through biochemical quantification of glycoprotein (GP) VI (immunoassay), heme (formic acid-based colorimetric assay), and DNA (dsDNA fluorescence assay kit) in thrombus homogenates [118]. Hemoglobin (ELISA) and heme content were highly correlated with RBC content determined by flow cytometry [119]. Different lymphocyte profiles were identified in cell suspensions of clots subjected to flow cytometry [120].
The presence of NETs was confirmed in intact thrombus samples subjected to endonuclease treatment to release NE activity [95], and by incubating with DNase-I to produce ex vivo thrombolysis [71]. Thrombin elution pattern assessed by measuring secreted thrombin activity along serial washings has been suggested as a biomarker of clot content [121].
Relationship between thrombus features and stroke etiology
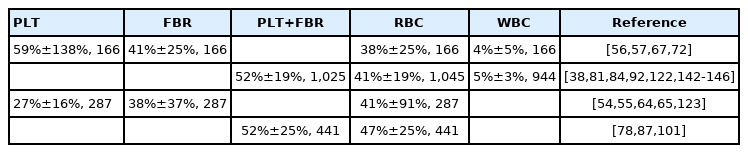
Analysis of thrombus size, structure and composition in patient cohorts found associations with TOAST etiology (large-artery atherosclerosis [LAA; TOAST 1]; cardioembolism [CE; TOAST 2]; stroke of other determined cause [ODC; TOAST 4]; cryptogenic [CRY] stroke of undetermined etiology [TOAST 5]), embolic stroke of undetermined source (ESUS), or underlying pathologies (Supplementary Table 2). LAA thrombi showed a larger ECA [39,40] and higher number of fragments [40]. Eight studies, reporting results from 1,183 thrombi (median 73), showed higher RBC proportions in thrombi from LAA or non-CE (LAA+ODC) sources [39,57,64,82,96,122-124]. Contrastingly, three studies (119 thrombi, median 37) showed higher RBC proportions in CE thrombi [72,84,125]. Seven studies (1,061 thrombi, median 58) showed higher FBR proportions in CE [39,57,76,82,96,123,124], while in two studies (82 thrombi, median 41) the FBR proportion was higher in LAA [72,125]. CE thrombi also showed a higher FP (FBR+PLTs) proportion in one study (137 thrombi) [122]. PLT proportion was higher in LAA in two studies (1,127 thrombi, median 563.5) [50,59], but higher in CE in another two studies (697 thrombi, median 348.5) [39,67]. Further support for higher PLT proportions in non-CE thrombi came from higher GP VI content in thrombus homogenates [118]. LAA thrombi showed mostly peripheral PLT distribution patterns (PDPs), while mostly clustering PDP was observed in CE [51]. Non-CE thrombi showed higher RBC/PLT ratio [73], in line with lower FP/RBC ratio [126]. Results from the large multicentric STRIP registry (1,350 thrombi), published during the preparation of this manuscript, showed that LAA thrombi had a higher mean RBC density (46%±23% vs. 42%±22%, P=0.01) and a lower PLT density (24%±18% vs. 27%±18%, P=0.03) than CE thrombi [147]. Regarding WBCs, three studies (358 thrombi, median 137) showed higher proportions in CE [89,96,122], while one study (37 thrombi) reported higher proportions in LAA [84]. Another study supported higher WBC proportions in CE, as estimated by DNA content in thrombus homogenates [118]. When WBC subtypes were analyzed, CE thrombi showed higher contents of neutrophils [76], NETs [66,93], and netting neutrophils [66], as well as more macrophages [67]. As for lymphocytes, CE thrombi contained more suppressor-cytotoxic T-cells [120], while LAA thrombi showed higher T-cell [90], helper T-cell, and natural killer (NK)-cell contents [120]. Finally, higher IL-1β expression was measured in LAA thrombi [112], while coagulation factor XIII was associated with CE [115].
Some studies aimed to compare the features of CRY thrombi with those of known TOAST etiology. Most of them found similar proportions of RBCs [39,57,64,96,122-124,127], FBR, PLTs (or both together) [57,76,96,122-124,127], and WBCs between CRY and CE thrombi [96,122,127]. CRY and CE thrombi also shared smaller ECA and number of retrieved fragments [39,40], clustering PDP [51], higher neutrophil counts [76], and NETosis [66], temporal profile of eluted thrombin activity [121], and low expression of IL-1β [112]. ESUS and CE thrombi showed similar RBC/PLTs ratios [73]. Conversely, a few studies found similar proportions of RBCs [84], PLTs [50], and WBCs [84,89] in CRY and LAA thrombi. Finally, ESUS and LAA thrombi shared low macrophage proportions [67].
Regardless of TOAST etiology, some thrombus features have been associated with the patient’s age and underlying pathologies. The clots from elderly subjects had higher FBR proportions compared to younger patients [128]. As for gross appearance, white thrombi were much more frequent in the context of pathologies like active cancer (AC) or infective endocarditis (IE) [36,38]. Furthermore, thrombi showed lower RBC proportions, and higher FBR and/or PLT proportions, with underlying AC [38,65] or diabetes mellitus (DM) [55]. Other composition characteristics were lower WBC proportion in AC [38], and higher NETosis in DM [94]. Direct presence of tumor cells in the thrombus was rare in AC [129]. Contrastingly, bacteria or fungi were present with underlying IE [7,109] or other systemic infections [109].
Thrombus analysis in individual cases was a valuable complementary tool in diagnosing the stroke etiology, especially atypical ones (Supplementary Table 3). Proteomic analysis determined a common origin for tandem occlusions after traumatic carotid thrombosis and embolization [130]. Macroscopic and thorough histopathological examination confirmed the occlusion by an intracranial atherosclerotic plaque, revealing the atherothrombotic etiology [19]. Identification of cocci or bacilli in the thrombus confirmed septic emboli in cases of bacterial IE [14,18,31,32,102,131-135], including a rare case of Whipple’s endocarditis [26]. Similarly, the presence of Candida confirmed septic embolus in a case of fungal IE [136]. Observation of fungal hyphae confirmed cases of septic embolus [137] and angioinvasive mucormycosis secondary to sinusitis [138]. Identification of papillary fronds or myxomatous tissue helped diagnose embolization secondary to cardiac papillary fibroelastoma [15,16,20] and myxoma [29], respectively. Visual assessment and histopathology identified valve tissue [24], calcifications [27,30], chordae tendineae [21], and aortic wall tissue [23] as embolism sources, which detached spontaneously or periprocedurally during valve replacement surgery. Immunohistopathology contributed to diagnose the embolism of a Libman-Sacks vegetation in systemic lupus erythematosus-associated endocarditis [22]. Thrombus calcification, cholesterol crystal cleft and foamy cells confirmed the aortogenic embolic stroke due to atheromatous lesion in the aortic arch [139,140]. Embolizations of carotid free-floating and carotid web thrombi were determined by assessing the clot aspect and blood cell content [28,141]. Clot visual appearance and high FBR content indicated thromboembolism secondary to coronavirus disease 2019-associated hypercoagulability [34] and cancer-related Trousseau syndrome [33]. Finally, thrombus examination confirmed periprocedural catheter-related thromboembolism during valve implantation [17] and inadvertent embolization of foreign bodies during aneurysm treatment [25].
Discussion
In response to the four research questions posed in this study, our results show that:
(1) Thrombi were subjected to physical analyses (macroscopic appearance, mechanical behavior, MRI, and synchrotron-based imaging), histopathological analyses (conventional histology, immunohistochemistry, and electron microscopy), biological analyses (biochemical, biomolecular, and cellular techniques, such as enzymatic assay, ELISA, transcriptomics, proteomics, metabolomics, and flow cytometry), and microbiological analyses (culture, histological staining, and metagenomics).
(2) The information obtained about thrombi included gross appearance (size, shape, consistency, visual texture, color, and homo/heterogeneous aspect), mechanical properties (stiffness, elasticity, tensile strength, and response to stress), structure (compactness, molecular and cellular organization, and ultrastructure), and composition (FBR, other coagulation factors, blood formed elements, NETs, vessel wall and plaque components, microbial pathogens, inflammatory mediators, protein cargo, metabolites, elements, and even foreign bodies).
(3) There were associations of thrombus size, structure and composition (mainly proportions of FBR and blood formed elements) with TOAST etiology and underlying pathologies, and similarities between cryptogenic thrombi and those of known TOAST etiology.
(4) Individual thrombus analysis proved to be a valuable complementary tool in the diagnosis of stroke etiology, particularly in atypical cases.
The first endovascular device clearance by the U.S. Food and Drug Administration occurred in 2004 [148], and the first report describing a systematic histological analysis of thrombi retrieved from cerebral arteries was published in 2006 [7]. Since then, a total of 134 eligible studies have been identified in this systematic review. Of note, there has been a surge of reports in the last 5 years, just after the publication of Brinjikji’s systematic review [3]. Although most studies carried out histopathological analyses, studies reporting biochemical or biomolecular analyses have been increasing in the last 3 years. Almost 10,000 thrombi/emboli retrieved from LVO stroke patients have been analyzed predominantly in cohort studies but also in case reports.
Physical, histopathological and biological analyses provided a considerable amount of information about the gross appearance, mechanical properties, structure, and composition of thrombi in patient cohorts. However, assessments of possible associations of thrombus features with stroke causes have been carried out mainly based on thrombus size, color, and proportions of FBR and blood formed elements. LAA thrombi were larger and more fragmented. Regarding composition, non-unanimous evidence supported higher RBC contents in thrombi from LAA or non-CE (LAA+ODC) sources, and higher FBR contents in thrombi of CE origin. WBC content, neutrophil count, and NETosis were also higher in CE thrombi. As for the PLT proportion, controversial histopathological evidence did not support a clear association with the CE or non-CE cause, although non-CE thrombi showed higher GP VI content (PLT marker), and peripheral PDP. Cryptogenic mechanisms account for 10% to 40% of all ischemic strokes [149]. Most evidence pointed to similar features in CRY and CE thrombi, mainly regarding proportions of RBCs, FBR, PLTs, and WBCs, and smaller size and number of retrieved fragments, clustering PDP, and higher NETosis. This could aid the etiologic investigation and reduce the percentage of CRY strokes. Of note, the large multicentric STRIP registry found statistically significant but clinically insignificant differences between clots of CE and LAA etiologies [147]. Although LAA clots had a higher mean RBC density and a lower mean PLT density than CE clots, the receiver operating characteristics analysis showed that identification of a reliable threshold with a high area under the curve for differentiating clots of these two etiologies based on composition analysis alone was not possible; this suggests that conventional histological analyses of the cellular composition do not provide insights into stroke etiology in cryptogenic cases. Regardless of TOAST etiology, a few studies found associations of thrombus color and composition with underlying pathologies like cancer, diabetes, and IE.
Macroscopic, histopathological, biomolecular, and microbiological analyses of thrombi were useful in the diagnosis of the stroke cause in individual cases. Thrombus features confirmed cases of typical intracranial and carotid thromboembolism, and atypical free-floating and web thrombi. Histopathology also contributed to diagnose embolisms due to cardiac tumor fragments, sterile endocarditis vegetations, aortogenic lesions, and tissue fragments detached during valve replacement surgery. Microbiological analysis revealed septic emboli secondary to IE, allowing appropriate antibiotic therapy. Finally, thromboembolisms due to secondary hypercoagulable states were diagnosed based on clot features.
Current methods of assessing thrombus features vary widely. Despite a consensus statement on the analyses of thrombi in AIS [1] and recommendations for thrombus handling and procedures [13], the results in this review show a lack of standardization in the research and reporting of thrombus characteristics and parameters. Heterogeneity in study designs, outcome measuring, and reporting precluded a meta-analysis of the association between thrombus composition and stroke etiology. Hopefully, ongoing multi-institutional registers, larger cohorts, and homogenous protocols will overcome this limitation. Although traditional histopathological techniques seem of limited value, the application of pathophysiological classifications show promise in differentiating between CE and LAA emboli [150]. Nevertheless, focusing on immunohistochemical analysis and more advanced techniques could help increase the knowledge on the composition and structure of thrombi. Advanced analysis of microRNA signatures [151], proteomic analysis [152], and combined “omic” analysis (proteome and metabolome) [153] are promising molecular approaches to elucidate the composition of emboli. Moreover, ultrastructural analysis using high-resolution SEM shows the finely organized clot components [56]. The role of all these advanced techniques in identifying stroke etiology could be further explored.
Conclusions
Although cohort studies report an abundance of quantitative rates of main thrombus components, a definite clot signature for accurate diagnosis of stroke etiology is still lacking. Nevertheless, qualitative examination of the embolus remains an invaluable tool in the diagnostic work-up of individual cases, particularly regarding atypical stroke causes. Beyond conventional histopathological and immunohistochemical clot analyses, future studies should emphasize the analysis of biomolecular composition and structural organization to provide insights about reliable links between clot features and stroke etiology.
Supplementary materials
Supplementary materials related to this article can be found online at https://doi.org/10.5853/jos.2021.02306.
Studies included in the qualitative synthesis
Cohort studies: thrombus composition and TOAST stroke etiology or underlying pathology
Case reports: thrombus/embolus composition and stroke etiologies or underlying pathologies
Notes
Disclosure
The authors have no financial conflicts of interest.
Acknowledgements
This work was partially supported by RETICS research network INVICTUS+ from Spanish ‘Instituto de Salud Carlos III’ (co-financed with European Regional Development Fund), through grant RD16/0019/0008. The funding source had no further role in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
The authors acknowledge the help rendered by Marta Méndez Debaets in manuscript preparation and reference management.