Prolonged Ictal Aphasia Presenting as Clinical-Diffusion Mismatch in a Patient with Acute Ischemic Stroke
Article information
Dear Sir:
The presence of clinical-diffusion mismatch (CDM) in patients with acute ischemic stroke may represent the ischemic penumbra, which requires emergency reperfusion therapy to improve stroke outcomes. We report here our experience of treating a patient with acute ischemic stroke and CDM. Specifically, the patient had a small infarct in the left temporal cortex and presented with sensory aphasia, but did not require reperfusion therapy. Magnetic resonance angiography and perfusion imaging findings were normal. Further investigation revealed that the aphasia was associated with ictal symptoms. Ictal aphasia is a considerable cause of non-oligemic CDM, perfusion imaging and angiographic studies may help discriminate true ischemic penumbra from non-oligemic CDM.
A 44-year-old, right-handed male patient presented with sudden onset language disturbance and headache for 24 hours. Approximately 30 years prior, the patient had undergone surgery to removal a right cerebellar tumor, which resulted in a history of mild dysarthria, hearing difficulties, right-sided ataxia, left-sided facial palsy, and right exotropia. There was no history of tumor recurrence or seizure. Neurologic examination revealed Wernicke's aphasia in addition to the preexisting neurological deficits, with an initial score on the National Institutes of Health Stroke Scale of seven. Diffusion-weighted magnetic resonance imaging of the brain revealed a small, focal, and high-signal intensity (volume 1.3 mL) with the apparent diffusion coefficient restriction in the left temporal lobe, compatible with acute infarction (Figure 1). Because there were no lesions that could explain the patient's aphasia, we diagnosed the condition to be significant neurological deficit with CDM and considered emergency revascularization. However, the revascularization procedure was not performed because we did not note any abnormal findings on magnetic resonance angiography or perfusion imaging.
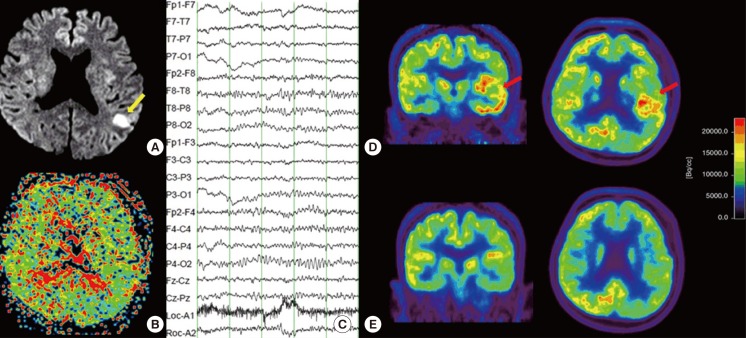
Representative images from magnetic resonance imaging and Fluorodeoxyglucose positron emission tomography (FDG-PET) of the brain. (A) Diffusion-weighted image showing a small cortical infarct (yellow arrow) in the territory of the left middle cerebral artery. (B) Perfusion image showing no obvious abnormalities. (C) Electroencephalography showing intermittent theta slowing in the left temporal cortex. (D) Images of FDG-PET, which was performed at the onset of symptoms, showing hypermetabolism (red arrow) in the left temporal cortex (coronal and axial view). (E) Images of the regions shown in D obtained during follow-up FDG-PET, which was performed when the symptoms resolved; they reveal normalization of left temporal hypermetabolism (coronal and axial view).
The results of the Korean version of the Western Aphasia Battery indicated severe Wernicke's aphasia (10/20 fluency, 1.9/10 auditory comprehension, 0.4/10 repetition, 0.5/10 naming, and 45.6/100 aphasia quotient). There were no abnormal laboratory results except a mild elevation in erythrocyte sedimentation rate (23 mm/h). The electrocardiogram showed a normal sinus rhythm, and chest x-ray showed no active lesion in either lung fields. During hospitalization, the severity of aphasia and mild confusion fluctuated. Electroencephalography demonstrated intermittent theta slowing in the left temporal areas (Figure 1). Fluorodeoxyglucose positron emission tomography (FDG-PET) showed hypermetabolism in the left temporal cortex (Figure 1). The aphasia was due to ictal and postictal symptoms following stroke rather than hypoperfusion-related symptoms. The patient was administered 250 mg/day phenytoin on the fourth day of admission. Symptoms remained on the following day, albeit attenuated; therefore, 1,000 mg/day levetiracetam was added to the treatment regimen. Symptoms resolved on day 8 of admission; however, the patient complained of dizziness and depressive mood. We therefore changed his medication at discharge to include 600 mg/day valproic acid, which was maintained for eight months. The patient has been seizure-free for one year (Figure 2), and the temporal hypermetabolism noted on FDG-PET has improved.
The predominant cause of sudden onset, prolonged aphasia, is stroke affecting the language network. In the present case, the patient developed sudden onset Wernicke's aphasia, with a small cortical high-signal intensity in the territory of the left middle cerebral artery on magnetic resonance imaging. Because the lesion did not explain the patient's symptoms, the aphasia was initially considered a symptom resulting from hypoperfusion in the territory of the left middle cerebral artery, and was therefore considered CDM. However, the cause of aphasia was subsequently thought to be an ictal and postictal symptom caused by acute ischemic stroke, due to the following reasons: First, there were no steno-occlusive lesions on magnetic resonance angiography or perfusion defects, even though the symptoms lasted more than two days. Second, the small cortical lesion itself could not explain the intermittent slowing in the left temporal area found on electroencephalography. Third, FDG-PET performed when the aphasic symptoms presented, showed hypermetabolism in the left temporal lobe. Fourth, aphasic symptoms and hypermetabolism on FDG-PET disappeared after the administration of antiepileptic drugs. Due to its low prevalence, the diagnosis of ictal aphasia is difficult, and only 56% of patients with aphasic seizures demonstrate electrographic ictal discharge on the first routine surface electroencephalography.1 FDG-PET imaging was useful for the diagnosis of aphasic nonconvulsive status epilepticus, although the electroencephalography findings were inconclusive.2
According to previous studies, CDM is associated with perfusion-diffusion mismatch, infarction expansion, and early neurological deterioration.3,4 It is therefore important to consider CDM when determining acute stroke management, such as thrombolysis or endovascular treatment.3,4 However, the false-positive diagnosis of ischemic stroke, labeled "stroke mimics", ranges between 1.3-25%, and seizures (38%) are the most common cause of stroke mimics.5,6 Moreover, acute ischemic stroke is important in the etiology of symptomatic seizures.
Nonconvulsive seizures can be confused with CDM, as seen in the case reported here. Initially, revascularization was considered as a treatment for CDM. However, CDM can also be due to stroke mimics, indicating that clinicians should consider the possibility of post-stroke seizures. Perfusion imaging, magnetic resonance angiography and FDG-PET can provide useful information for differentiating non-oligemic CDM from true ischemic penumbra.
Notes
This study was supported by a grant from the Korea Healthcare Technology R&D Project, Ministry of Health and Welfare Republic of Korea (HI10C2020).
The authors have no financial conflicts of interest.
