Lack of Association of Clinical Factors (SAMe-TT2R2) with CYP2C9/VKORC1 Genotype and Anticoagulation Control Quality
Article information
Abstract
Background and Purpose
Advantages of new oral anticoagulations may be greater in atrial fibrillation (AF) patients of poor anticoagulation control with warfarin. The SAMe-TT2R2 scoring system, based on clinical variables, was recently developed to aid in identifying these patients. In this study, we investigated the association of this clinical composite score with genetic factors related warfarin dosing and the quality of anticoagulation control.
Methods
Clinical and genetic data were collected from 380 consecutive Korean patients with AF (CHA2DS2-VASc score, 3.5±1.8) who were followed for an average of 4 years. We evaluated factors associated with time in therapeutic range (TTR, INR 2-3), including the CYP2C9 and VKORC1 genotypes and the SAMe-TT2R2 score (Sex female, Age <60 years, Medical history [>two co-morbidities], Treatment [interacting drugs, e.g., amiodarone], Tobacco use within 2 years [doubled], and Race non-white [doubled]).
Results
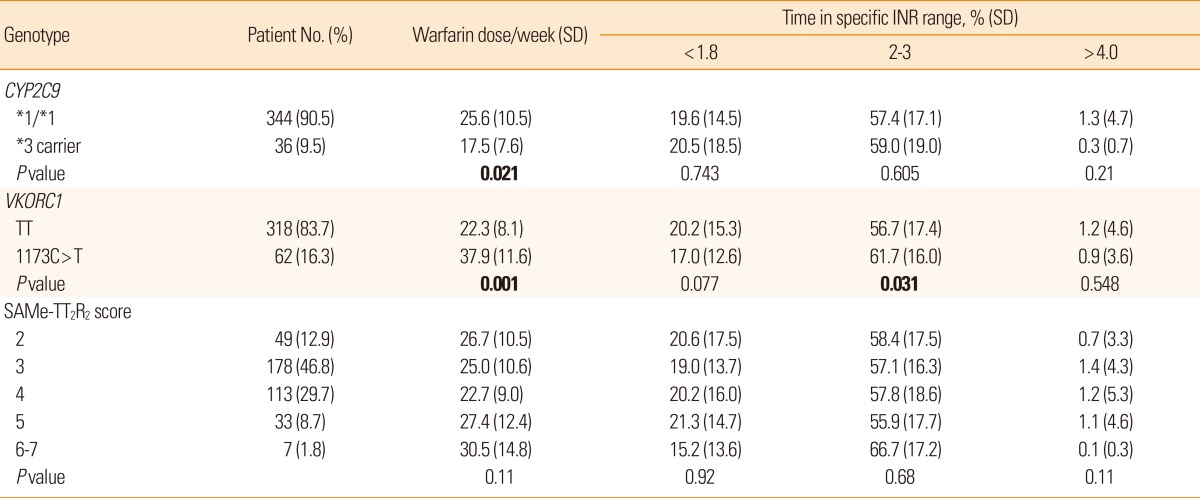
The average SAMe-TT2R2 score was 3.4±0.9, range 2-7; and 153 patients (40.2%) had SAMe-TT2R2 scores ≥4. Time in specific INR ranges varied depending on the VKORC1 genotype but not with the CYP2C9 genotype or the SAMe-TT2R2 score. TTR was higher in patients with the VKORC1 1173C>T than in VKORC1 TT (61.7±16% vs. 56.7±17.4%, P=0.031). Multivariate testing showed that VKORC1 genotype but not the SAMe-TT2R2 score was significantly associated with labile INRs. There was no correlation between the SAMe-TT2R2 scores and pharmacogenetic data.
Conclusions
A genetic factor, but none of the common clinical and demographic factors, as combined in the SAMe-TT2R2 score, was associated with the quality of anticoagulation control in Korean patients with AF.
Introduction
Although anticoagulation with the use of warfarin may effectively reduce risk of cardioembolic stroke in patients with atrial fibrillation (AF), warfarin has a narrow therapeutic window. In the RE-LY trial with carefully selected and monitored patients, the proportion of time in therapeutic range (TTR), defined by the international normalized ratio (INR) of 2 to 3, varied from 44% to 77% depending on the study or clinical center.1 Hemorrhage or thrombosis due to over- or under-dosing may have devastating consequences. Poor coagulation control may increase the risk of thromboembolic events2 and warfarin-related bleeding,3 and influence the size of brain infarcts.4 Also recent study shows the importance of identification of the patient with atrial fibrillation who has higher risk of stroke, and administration of proper anticoagulation.5
New oral anticoagulants (NOACs), i.e., dabigatran, apixaban, rivaroxaban, and edoxaban,6,7,8,9 are not inferior to warfarin in preventing ischemic stroke and systemic embolism in patients with non-valvular AF, and are superior to warfarin in reducing hemorrhagic stroke. For all vascular events, non-hemorrhagic events, and mortality, advantages of NOACs may be greater in the case of poor INR control than with good INR control.1 However, no consensus exists regarding the indication for use of these agents in patients with AF.
The quality of anticoagulation control may depend on genetic factors, notably CYP2C9 and VKORC1 polymorphisms, and on non-genetic patient-related factors, such as gender, race/ethnicity, and paroxysmal vs. permanent AF. Dr. Lip and colleagues recently introduced the SAMe-TT2R2 score, a validated assessment scheme based on clinical variables, to aid in distinguishing patients with AF who are likely to do well on warfarin (having low SAMe-TT2R2 scores) from those who are likely to have poor anticoagulation control (predicted by high scores).10 This score appears to provide valuable information relevant to safety and effectiveness of treatment, while avoiding the time and expense of pharmacogenetic study.11 However, the SAMe-TT2R2 score was derived from clinical trial data and did not include all of the consecutive INR measurements.
Thus, in the present investigation we tested both the SAMe-TT2R2 score and pharmacogenetic data for power to predict anticoagulation control, expressed as the TTR, and the correlation between the two predictors. Data for analysis were retrieved retrospectively and included the consecutive INR measurements as well as the CYP2C9 and VKORC1 genotypes for each patient.
Methods
Patient selection
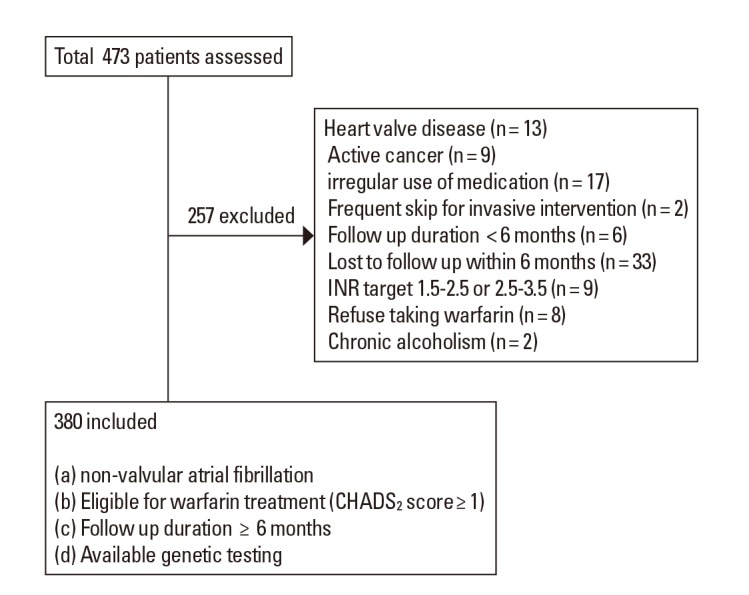
We collected data from consecutive patients who visited a University Medical Center, Seoul, Korea. Patients included in this study met the following criteria (a) diagnosis of non-valvular AF, (b) eligibility for warfarin treatment to prevent thromboembolism (i.e. CHADS2 score ≥1), (c) genetic testing data available, and (d) follow-up greater than 6 months. Patients were excluded for (a) irregular use of medications (>20% missed doses or >10% extra doses),12 (b) active cancer (c) chronic alcoholism, and (d) chronic consumption of food, beverages, or medications that may interfere with warfarin metabolism, such as green liquor and herbal medications.13,14,15 Patients with a target INR range of 1.5 to 2.5 or 2.5 to 3.5 were also excluded. Those taking antiplatelet in addition to anticoagulant medication were not excluded from this study. The Institutional Review Board approved this study and all participants gave informed consent before the study began. Patient selection is shown in Figure 1.
Work-up
Patients received long-term warfarin treatment with a target INR range of 2.0 to 3.0. After stabilization, maintenance dosing was achieved by performing periodic INRs. Dose assessment was performed every 1 to 3 months. Following enrollment of a patient, we obtained a medical history, performed a physical examination, and drew blood for INR measurement and CYP2C9 and VKORC1 genotyping. We contacted each patient as scheduled for an INR test, to assess warfarin dosing, and to ask about the use of any new medications. Compliance with warfarin treatment was regularly monitored and patients received dietary instructions for use with warfarin therapy.
Variables that could potentially affect the quality of anticoagulation control were recorded for each patient. These included age, body surface area, concurrent medications, and risk scores for thrombosis (CHA2DS2-VASc)16 and hemorrhage (ATRIA)17. In this study, we used the ATRIA score for bleeding risk instead of the HAS-BLED score because a labile INR is a key component of the HAS-BLED risk scheme.3 The methods used for CYP2C9 and VKORC1 genotyping are explained in our previous publications.18,19
The SAMe-TT2R2 scores were determined for each patient (Sex female, Age <60 years, Medical history [more than 2 comorbidities], Treatment [interacting drugs, e.g., amiodarone for rhythm control], Tobacco use within 2 years [doubled], Race nonwhite [doubled]). These factors are reportedly associated with poor control of INR in AF patients on warfarin therapy; that is, the factors predict a low value of TTR.10 Medical history was defined as having more than two of the following: hypertension, diabetes, coronary artery disease/myocardial infarction, peripheral arterial disease, congestive heart failure, previous stroke, pulmonary disease, and hepatic or renal disease.10 In this study, all participants were Korean.
Quality of anticoagulation control
To calculate the proportion of time spent within specific INR levels, Rosendaal's method was used.20 To estimate the unknown INR values (INRx) between two sequential INR values, linear interpolation of INR was performed.

We evaluated the relationship between CYP2C9 and VKORC1 genotype and TTR (defined as 2.0-3.0 for all patients) and measured times within specific ranges of INR. These ranges were less than 1.8, over 4.0, and between 2.0 and 3.0. INR levels after one month of initiation of warfarin were used to define quality of anticoagulation control based on detection of labile INRs during warfarin maintenance. In this analysis, we excluded INRs obtained during temporary discontinuation, and during the first month after treatment was restarted. We also evaluated the relationship between the SAMe-TT2R2 scores and TTR, and the relationships of the SAMe-TT2R2 scores with CYP2C9 and VKORC1 genotypes.
Statistical analysis
All numeric data are presented as the mean±SD or median (interquartile range [IQR]). Differences in clinical and laboratory parameters were evaluated by one-way ANOVA with post hoc analysis using the Fisher's least-square difference or Kruskall-Wallis test for continuous variables and Pearson's chi-square, Fisher's exact test or linear by linear association for categorical variables. An independent t test or the Mann- Whitney U test was used to evaluate differences in factors between groups. Independent factors for a patient having labile INRs were evaluated using logistic regression. In this study, a TTR of <65% was taken to indicate a labile INR and ≥65% as stable INR. The Institute for Health and Care Excellence (NICE) commissioning guide recommended that TTR of over 65% are desirable in the anticoagulation control.21 In the present study, 35% of patients had TTR ≥65% and 65% had TTR <65%. Among patients with TTR <66%, the NOAC are reported to reduce risk of major bleeding more effectively relative to warfarin than in patients with TTR ≥66%.22 Body surface area, warfarin dose and dose assessment interval, concurrent medications, liver disease, CHA2DS2-VASc and ATRIA scores, the SAMe-TT2R2 score, and CYP2C9 and VKORC1 genotype all served as predictive variables. All variables with P<0.20 in the univariate analysis and variables of interest in this study (e.g., SAMe-TT2R2 score) were included in the multivariate analysis. In the final models, differences with P values<0.05 were considered to be significant. All statistical analyses were performed using SPSS for Windows, Version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
Patients' characteristics
Clinical and genetic data form 380 Korean patients with AF were collected and the patients were followed for about 4 years (mean±SD, 231.9±199.7 weeks). Mean age was 70.6 (ranging from 22 to 90 years) and 153 patients were female. The average CHA2DS2-VASc score was 3.51±1.74, and the ATRIA score was 2.23±2.17. Blood sampling for warfarin dose assessment was performed at a mean±SD interval of 5.5±2.8 weeks.
The SAMe-TT2R2 score was 3.4±0.9, range 2-7, and 153 subjects (40.2%) had SAMe-TT2R2 scores of ≥4. No scores of 0 or 1 were found in this study because the risk scheme assigns two points for non-white race classification. Genotype frequencies for CYP2C9*3 (rs1057910) and VKORC1 1173C>T (rs9934438) were 9.5% and 16.3%, respectively. Clinical, genetic and demographic characteristics of the patients are summarized in Table 1.
Factors associated with quality of anticoagulation control
For the patient group overall, irrespective of genotype, the percentage of time spent in therapeutic range was 57.5% during the entire follow-up period. Time in specific INR ranges showed dependence on VKORC1 genotype (Table 2). Carriers of VKORC1 TT spent less time in therapeutic range (INR 2-3) than carriers of VKORC1 1173C>T (P=0.031). On the contrary, the percentages of time in specific INR ranges did not differ according to CYP2C9 isoform or SAMe-TT2R2 score.
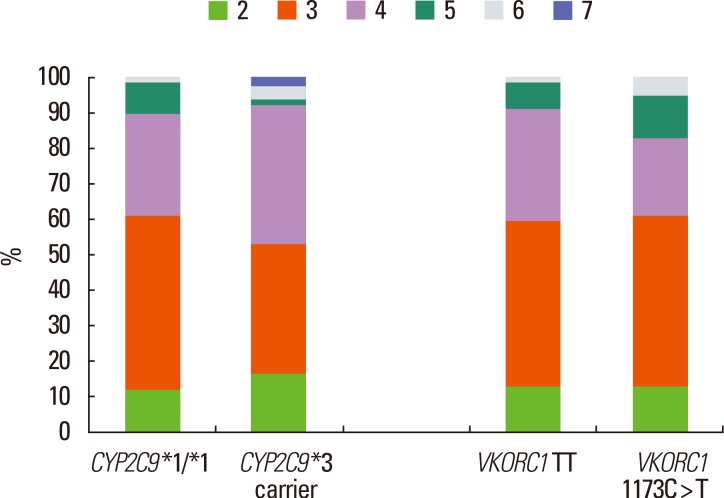
The clinical score and pharmacogenetic data did not correlate well. We expected a negative correlation between the SAMe-TT2R2 score and time in therapeutic range (INR 2-3), but the Spearman correlation analysis showed no correlation (r=0.11 P=0.826). The distribution of the SAMe-TT2R2 scores was similar among the different genotypes related to warfarin dosing (Figure 2).
Multivariate testing was performed to test further for factors that may independently predict labile INR (Table 3). The VKORC1 TT but none of the other factors, including the SAMe-TT2R2 score, was significantly associated with labile INR.
Discussion
The main findings of this study were that (1) a genetic factor related to warfarin response, but no common clinical or demographic factor, including the SAMe-TT2R2 score, was associated with the quality of anticoagulation control, and (2) there is poor correlation between clinical and genetic factors.
The American Heart and Stroke Association guidelines for oral antithrombotic agents for the prevention of stroke in non-valvular AF23 support the use of both warfarin and NOACs to prevent first and recurrent strokes in patients with non-valvular AF. The guidelines recommend the individualized selection of antithrombotic agent(s) based on risk calculations, cost, tolerability, patient preference, potential for drug interactions, and other clinical characteristics, including TTR if the patient has been taking warfarin. In determining the selection of warfarin vs. NOACs, a dilemma arises in that the TTR cannot be known at the time of initial treatment. As a result, it is difficult to identify the newly diagnosed patients with AF who would do well on warfarin and who also have high values for time in therapeutic range. This is desirable because the main benefits of NOACs compared with warfarin may be only marginal in those with high times in therapeutic range, although the reduction in intracranial hemorrhage is still evident.24 In a recent meta-analysis with data for four phase 3 clinical trials of NOACs, the NOACs had a favorable risk-benefit profile as compared with warfarin that was consistent across diverse patient groups.22 However, it also showed a greater relative reduction in major bleeding with NOACs when TTR was less than 66% than when it was more than 66%.22 High TTR (>70%) are known to be associated with the best efficacy and safety of warfarin.25
Genotype-guided dosing of warfarin was recently reported to be associated with a higher percentage of time in the therapeutic INR range than standard dosing during the initiation of warfarin therapy.26 However, the relationship of warfarin pharmacogenetics to long-term TTR during warfarin maintenance is not well studied. If pharmacogenetic analysis could predict TTR at the time of the initial AF diagnosis, the selection of antithrombotic agent might be clarified. In particular, those patients who have genetic variability associated with labile INR during the maintenance of warfarin could be selected for treatment with NOACs. Our results showed, for example, that patients with VKORC1 1173C>T frequently show poor anticoagulation control. However, because polymorphism frequency varies between ethnic groups, the results of this study may not apply to other populations with different distributions of CYP2C9 and VKORC1 isoforms.27 Further studies are needed to determine the clinical significance of warfarin pharmacogenetics in ethnic groups with higher prevalence of CYP2C9 variants.
Our findings, that clinical and demographic factors expressed in the SAMe-TT2R2 score were not associated with quality of anticoagulation control, contradict the recent findings of Dr. Lip and colleagues [9]. This contradiction may be explained in various ways. In the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM), the same patient cohort served in both the derivation and validation phases of the study. Although the mean age (~70 years-old) and mean duration of follow-up (3.5-3.9 years) were similar between that study and ours, the baseline patient characteristics differed significantly.28 In particular, most subjects (89%) in the AFFIRM study were white, whereas all patients in our study were Korean. "Non-white race" represents two points from a total of eight points in the SAMe-TT2R2 risk prediction scheme. In addition, only 13% of the AFFIRM study subjects, but 57% of those in our study had experienced stroke or TIA. The choice of NOACs vs. warfarin would be particularly important in patients who have suffered stroke resulting in severe disability, because the monitoring of INR is often difficult in these patients; and importantly, the cost of NOACs may be prohibitive in developing countries, which include a large proportion of the world's population. In this study, furthermore, patients with valvular AF (who are not candidates for NOAC treatment) and those with a target INR range of 1.5 to 2.5 or 2.5 to 3.5 (outside the INR target of 2-3) were excluded, whereas 11%-12% of both the derivation and validation groups in the AFFIRM study had valvular AF. Another important difference between our study and that of Lip et al., not all of the AFFIRM participants received anticoagulation therapy throughout the study follow-up period and consecutive INRs were not measured. Instead, patients with >12 months of uninterrupted warfarin treatment and more than eight INR values available were included in their analysis.10 Our results are in line with recent results from a Danish cohort. Recently, Jane Skov et al.29 investigated to determine whether the SAMe-TT2R2 score can predict INR control and the population only included whites, and showed that the SAMe-TT2R2 score was not predictive of TTR. Thus, results of our and other studies indicate that further studies are needed for external validation of SAMe TT2R2 score.
The strength of the present study resides in the consecutive recruitment of patients with comprehensive evaluation of factors related to the quality of anticoagulation control, such as concurrent medications, scored risk assessments for AF-related thromboembolic and hemorrhagic events, and consecutive INR measurements with exclusion of INR values during temporary treatment interruptions and during the first one month after treatment was resumed. Limitations of this study include the retrospective design and residence at a single clinical center. Although we cannot generalize our findings in Korean subjects to all other ethnic groups, our analytical approach and findings may inform clinical services for relatively large populations throughout Asia, and possibly Africa. Further prospective studies are needed to identify clinical and genetic factors related to the quality of anticoagulation control during treatment with warfarin in Asians.
Conclusion
In conclusion, genetic factors, but not the commonly recognized clinical and demographic factors, were associated with quality of anticoagulation control in patients of Asian background who had recently survived an AF-related stroke. Our findings emphasize the need for continuous efforts to find clinical scoring systems to reliably inform therapeutic decisions, such as the choice between warfarin and less widely available NOACs. In addition, further studies are needed to test whether NOACs may be particularly effective in patients who have genotypes related to INR lability during warfarin maintenance.
Notes
This study was supported by the Korean Healthcare Technology R&D Project, Ministry of Health & Welfare (A110208).
The authors have no financial conflicts of interest.