Introduction
Moyamoya disease (MMD), also called spontaneous occlusion of the circle of Willis, is a chronic, occlusive cerebrovascular disease of unknown etiology characterized by steno-occlusive changes at the terminal portion of the internal carotid artery (ICA) and an abnormal vascular network at the base of the brain [1]. In light of the increasing number of patients with unilateral involvement [2] as well as evidence that a substantial number of unilateral cases progress to bilateral presentation [3,4], the diagnostic criteria for definitive MMD were revised to include patients with both bilateral and unilateral presentation of terminal ICA stenosis with an abnormal vascular network at the base of the brain (statement by the Research Committee of MMD of the Japanese Ministry of Health, Labour, and Welfare in 2015). Diagnostic criteria also state that definitive diagnosis of MMD requires catheter angiography in unilateral cases, whereas bilateral cases can be promptly diagnosed by either catheter angiography or magnetic resonance imaging/angiography (MRI/MRA). When there are causative diseases or associated conditions, terms such as moyamoya syndrome and angiographic moyamoya are often used [5]. Although uncommon, MMD is an important cause of non-atherosclerotic intracranial arterial disease, especially in East Asian countries. In these regions, isolated middle cerebral artery stenosis is seen in young patients, with eventual evolution into MMD [6]. MMD is the most important cause of stroke or transient ischemic attack (TIA) in children in this part of the world. Here, the epidemiology, clinical features, and diagnosis of MMD are reviewed.
Epidemiology
For an unknown reason, MMD is relatively common in people living in East Asian countries such as Korea and Japan, as compared to those in the Western Hemisphere. Table 1 summarizes epidemiological study results in East Asian countries. According to a survey performed in Japan in 1995, the prevalence of MMD was approximately 3.16/100,000, with an estimated incidence of 0.35/100,000 [7]. A subsequent survey in 2004 showed that the number of patients considerably increased, with an estimated incidence of 0.54/100,000, and prevalence of 6.03/100,000 in 2003 [8]. Another study performed in Hokkaido, Japan [8], showed that the incidence and prevalence were 0.94/100,000 and 10.5/100,000, respectively, based on 267 newly diagnosed MMD patients between 2002 and 2006. The male-to-female ratio was 1:1.8 or 1:2.2 in these surveys, and approximately 10%-15% of patients had a family history. The risk of having MMD in family members is about 30-40 times higher than the general population [7,9]. There were two peaks of incidence, at 10-20 and 35-50 years old.
An epidemiology study from Korea based on National Health Insurance (NHI) data [10] reported that the prevalence of MMD increased from 6.3/100,000 in 2004 to 9.1/100,000 in 2008. The incidence was 1.0/100,000 in 2008. Beginning in 2006, the NHI initiated a registration program for 138 rare diseases, including MMD. Patients registered in this program become eligible for a copayment reduction. According to a study using the data set obtained from 2007 to 2011 [11], the number of incident MMD cases increased from 848 to 1,192, with the annual incidence being 1.7 to 2.3/100,000. In both studies, the female-to-male ratio was 1.9, and there were two peaks of incidence; the peak in the pediatric group occurred at age 5 to 14 years, accounting for 16.2% of all incident cases, and the peak in the adult group occurred at age 45 to 54 years, accounting for 22.8% [11] (Figure 1). The overall prevalence of MMD in 2011 was as high as 16.1/100,000.
Although Korea has an apparently higher incidence and prevalence of MMD compared with Japan, it should be noted that a direct comparison is difficult for the following reasons. There were differences in data collection methods and population characteristics; Japanese studies used data from hospital surveys, with a limited response rate. On the other hand, Korean studies were based on national insurance data. Moreover, because registered patients were financially supported from 2006, there is a possibility of overdiagnosis. Additionally, the Japanese study analyzed data in 1995 and 2006, whereas the Korean studies analyzed data from 2004 to 2011. Thus, a progression of time may be the reason for the apparently higher prevalence in Korea.
Recently, the inpatient databases of the Taiwan NHI program identified MMD patients during 2000 to 2011 [12]. During the 12-year period, 422 patients were identified, representing an annual incidence of 0.15/100,000 person-years. Adults exhibited an upward trend, with an incidence rate of 1.74 in 2010 to 2011 compared with 2000 to 2001. Thus, the incidence of MMD has increased in adults but not in children from 2000 to 2011 in Taiwan. For unknown reasons, compared with patients hospitalized during 2000 to 2005, patients identified during 2006 to 2011 had a greater female-to-male ratio (1.7 vs. 1.1). Epidemiology studies in mainland China are rare. An epidemiological study in the area of Nanjing, the capital city of Jiangsu province, with a total population of approximately 6.2 million, showed that the prevalence was 3.92/100,000 during 2000-2007 [13], which was generally lower than that in Korea or Japan (Table 1). Unlike series from Korea and Japan, hemorrhage was more common than ischemic stroke in this cohort, probably due to the fact that the second peak (adult group) was larger than the first (pediatric group). However, this study was based on data from a small part of China. A more recent Chinese study on 802 MMD patients [14] showed that clinical presentations are similar to those from Korea and Japan. In both studies, age distribution of the patients with MMD had a bimodal occurrence: at age 5 to 9 years, and 35 to 39 years. Although reliable, national epidemiologic studies on mainland China are not yet available; given the lower incidence of MMD in the Taiwan study based on NHI data, the incidence of MMD in Chinese people may be lower than in Korean and Japanese counterparts. More surprisingly, unlike Korea and Japan, there is no difference in sex distribution of MMD in Chinese patients [13,14]. However, these studies do not represent data from all of China. Moreover, the female-to-male ratio increased over time from 1.1 (2000 to 2005) to 1.7 (2006-2011) in a Taiwan study [12]. Therefore, a conclusion should be cautiously drawn. In addition, familial occurrence of MMD has been variably reported: 1.5% [13] or 5.2% in China [14], and 10%-15% in Japan [7,8]. Although familial cases were previously (in 1997) reported to be low (2%) in South Korea [15], more recent studies showed that familial history was found in 10%-15% of MMD patients [16-18]. The ratio of familial cases may change according to the extensiveness of diagnostic workup; a recent Chinese study [19] screened 285 immediate family members of 245 sporadic MMD patients with transcranial Doppler (TCD) and identified 41 cases of familial MMD. Thus, the familial cases increased from 7% (before screening) to 15% (after screening). Because TCD has a high diagnostic agreement with MRA, the authors recommended implementation of TCD screening in all family members of MMD patients to detect asymptomatic cases. As shown in Table 1, there is an increasing incidence and prevalence of MMD over time in East Asian countries. This finding may indicate an actual increase in the incidence of MMD in these countries. However, a more plausible explanation would be an increase in newly diagnosed patients owing to the recent advent of noninvasive diagnostic tools such as MRA. Another explanation would be an increasing number of survivors due to improved management. Studies from outside of Asia are rare. In Washington state and California, the incidence of MMD was reported to be 0.086/100,000 based on 298 patients. The incidence was the highest in Asians, followed by Blacks, Whites, and Hispanics (Table 1). The incidence in Asian Americans was 4.6 times higher than that in Whites. Female preponderance was also noted [20]. African-Americans had an earlier disease onset with a median age of 18. However, a more recent study based on the Nationwide Inpatient Sample database reported that MMD appears to be distributed among the races according to their relative proportions in the USA population [21]. From 2005 to 2008, there were an estimated 7,473 (2,236 pediatric and 5,237 adult) patients admitted with a diagnosis of MMD in the USA. MMD patients were most frequently Caucasians. Overall, ischemic stroke was the most common reason for admission in both children and adults. Hemorrhagic stroke was more frequent in adults compared with children, and there was a bimodal age distribution with peaks in the first and fourth decades of life. Female-to-male ratio was 2.2. Thus, MMD in the USA does not seem to differ from East Asian MMD.
Clinical features
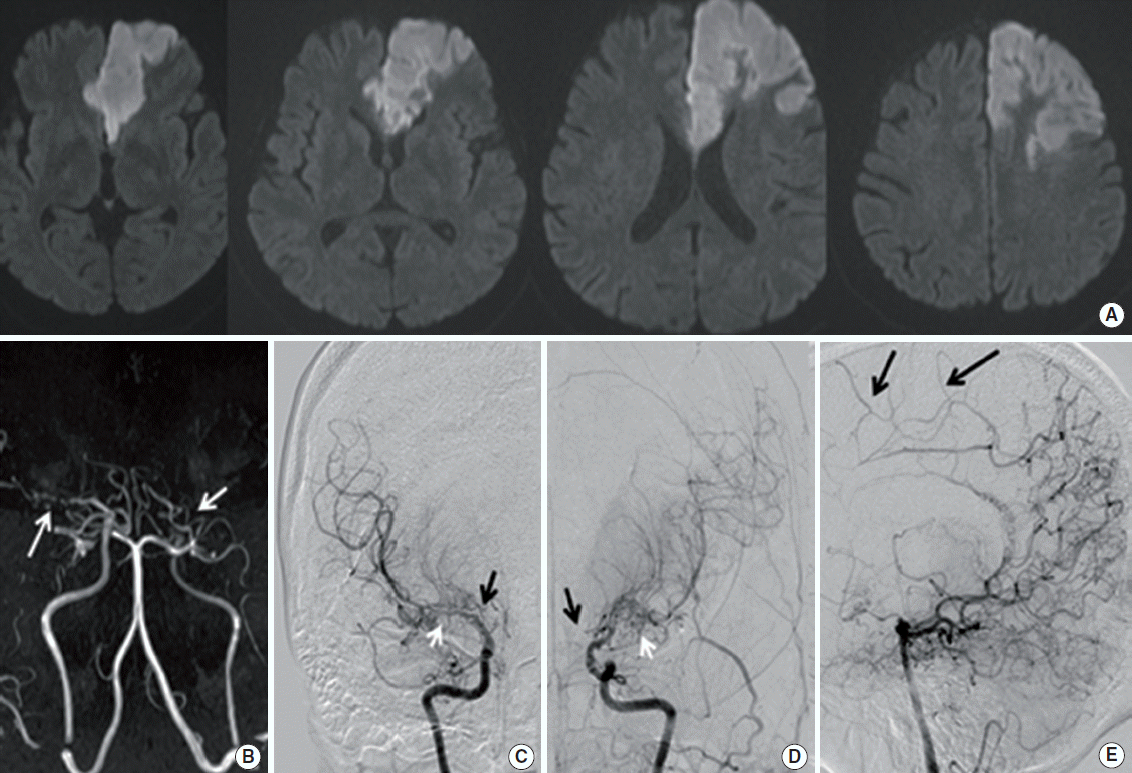
The clinical presentations of MMD include TIA, ischemic stroke, hemorrhagic stroke, seizures, headache, and cognitive impairment. The incidence of each symptom varies according to the age of the patients [22]. An ischemic event is the most important clinical manifestation of MMD. Cerebral hypoperfusion due to progressive major vessel occlusion results in repeated hemodynamic TIAs or ischemic strokes in children or young adults. Thus, MMD is a classic example of a hemodynamic cause of stroke. We often encounter patients who develop repeated TIAs when they are hyperventilating, crying, eating hot noodles, or playing a harmonica or flute. Decreased arterial PaCO2 due to hyperventilation may possibly induce vasodilation of normal vessels and subsequent hypoperfusion in vulnerable areas via a steal phenomenon. Stress, fatigue, infection, and dehydration may also precipitate ischemic symptoms. Less often, patients have territorial infarctions due to embolism or thrombotic occlusion in the distribution of the middle cerebral artery (MCA), anterior cerebral artery (ACA), or posterior cerebral artery (PCA) (Figures 2 and 3) [23]. The anterior circulation is predominantly involved, and the most frequent ischemic symptom is hemiparesis, followed by speech disturbances and hemisensory abnormalities [24]. In one study that examined 410 pediatric MMD cases, ischemic symptoms were attributed to MCA territory in 92, ACA territory in 52, and PCA territory in 10 [25]. Although posterior circulation stroke has been considered uncommon, developing in the late stage of MMD [26], a more recent study reported that PCA involvement was present in 29% of patients, with 17% demonstrating PCA territory infarction [27]. Unexpectedly, the prevalence of PCA involvement was not much different (26% vs. 33%) between pediatric and adult patients. Although PCA involvement is the natural disease process of MMD, given the observation that PCA disease progression frequently occurs after anterior circulation revascularization procedures, some argue that secondary stenosis (involution of the PCA) may also occur, owing to improved collaterals to the anterior circulation [28]. Regardless of the cause, PCA involvement is one of the factors related to poor prognosis in MMD [25]. Recently, a correlation between the homozygous c.14576G > A variant of the ring finger protein 213 and early onset and aggressive PCA in volvement has been shown [29], providing further evidence for the significance of PCA stenosis as a prognostic factor. In MMD patients, the infarct topography does not often fit in classic vascular territory (Figures 2 and 3). This is partly because of the concomitant presence of hemodynamic insufficiency and partly because of altered vascular territories secondary to long-standing major vessel occlusion, along with diversely developed collateral channels. In one study [30], the authors examined diffusion-weighted MRI patterns of 91 cerebral infarcts from 66 MMD patients. Each ischemic lesion was categorized as an unusual type (gyral, atypical territorial, and honeycomb patterns) or a typical (usually seen in stroke patients) type (territorial, multiple-dot, borderzone, and deep lacunar pattern). The infarct pattern was more often atypical (gyral, 44%, atypical territorial 13%, and honeycomb 11%) than typical (territorial 10%, multiple-dot 9%, borderzone 6 %, and deep lacunar 8%). Gyral and borderzone patterns were more frequently seen in pediatric (< 20 years old) MMD patients, whereas a honeycomb pattern was more common in adult patients with advanced vascular stages, which may be associated with age-specific vulnerability of the brain to ischemia, stage of arteriopathy, or changes of abnormal collateral pathways. Approximately 30% of MMD patients present with intracerebral hemorrhage (ICH) (or less commonly, subarachnoid hemorrhage) secondary to friable collateral vessels harboring microaneurysms or false aneurysms [24]. The hemorrhages usually occur in the anterior circulation territory. Although any brain region can be affected, intraventricular hemorrhage occurs more commonly than in hypertensive hemorrhage [31]. In one case-control study that compared 93 patients with ICH associated with MMD and those with primary ICH [32], the location of hemorrhage was significantly different between the two groups (Figure 4). The order of frequency of the location was putaminal (46.2%), thalamic (19.4%), pontine (14%), lobar (9.7%), cerebellar (4.3%), caudate (4.3%), and pure intraventricular (2.2%) in primary ICH, compared to intraventricular (37.6%), lobar (23.7%), and putaminal (22.6%) in MMD patients. ICH in the pons and cerebellum only occurred in primary ICH patients, whereas callosal hemorrhage (4.3%) only occurred in MMD-associated ICH. Primary intraventricular hemorrhage in MMD patients was closely associated with a prominent ipsilateral anterior choroidal artery, suggesting that rupture of focal microaneurysms in the abnormally dilated anterior choroidal artery branches may produce intraventricular hemorrhages. In addition, it has been shown that gradientecho T2* weighted or susceptibility-weighted MRI identify cerebral microbleeds (CMBs) in 28%-46% of MMD patients [33-35]. According to Sun et al. [35], CMBs most often occurred in deep and periventricular areas, and are associated with dilated anterior choroidal or posterior communicating arteries. They hypothesized that CMBs represent red blood cells leaked from these dilated, fragile vessels. Periventricular CMBs were associated with future occurrence of intraventricular hemorrhages. Thus, MMD should be suspected when the patients are young, without vascular risk factors, or when the location of the hemorrhage is atypical. For an unclear reason, hemorrhagic strokes are less common in Caucasians than in Asians [31,36,37].
With significant brain hypoperfusion, cognitive impairment, intellectual decline, or mental retardation may develop [38-40], which is a grave problem for children of school age. Seizures occur in approximately 5% of patients secondary to an ischemic lesion or hypoperfusion, usually starting in childhood. Headache either presents as a symptom of MMD or newly develops after bypass surgery. Although the prevalence of headache in MMD has been considered to be low [24], a recent study [41] reported that 44 of 204 (21.6%) children with MMD suffered from headache, and nausea/vomiting were seen in 12. In 4, headache developed during hyperventilation, and in 3, TIA and headache occurred simultaneously. The cause of headache in MMD remains unclear. Cerebral hypoperfusion may possibly lower the threshold for migraine development and increase the risk of spreading cortical depression [42,43]. The headache occasionally improves after the revascularization process, suggesting that cerebral hypoperfusion is related to the pathogenesis of headache at least in some of these patients. The experience of preoperative headache is a strong predictor of postoperative headache [41]. Although uncommon, MMD may present with involuntary movements, usually in children [44]; in one study, 17 of 410 (4%) pediatric MMD patients developed involuntary movements [25]. Baik and Lee [45] studied 42 patients (38 described in previous studies and 4 from their hospital) who developed involuntary movements associated with MMD. Twenty-seven patients developed chorea; 4, dystonia; 4, a mixture of both; and 7, dyskinesia. In 37 patients, brain imaging studies showed ischemic lesions, but the remaining 5 showed no parenchymal lesions. Studies showed cerebral hypoperfusion in the basal ganglia and in the cerebral cortical areas on the relevant side. In most of the patients, symptoms improved regardless of treatment.
There are two peaks of incidence with different clinical presentations, at around 10 years and 30-40 years, although one Japanese study reported that there are three peaks [9]. The peak appears to occur later in women than in men [9]. In children, ischemic symptoms, especially TIAs, are predominant (70%) [25,46]. Intellectual decline, seizures, and involuntary movements are also more common in this age group. In contrast, adult patients present with intracranial hemorrhage more often than pediatric patients. These different age peaks and clinical presentations in each age group were also observed in MMD patients living in the USA [20,21]. There have been debates about whether MMD in European Caucasians differs from that in Asians. A recent study on 153 European Caucasians with MMD [47] showed that although they also have two age peaks (11-18 and 40-49 years), the later peak was longer than in Asian patients. The later peak was associated with a stronger female preponderance (2.9:1). On the other hand, only 1.3% of cases were familial. Despite a relatively predominant adult patient population, hemorrhagic stroke overall occurred less frequently (8.5%) than in Asian patients, and occurred more frequently in pediatric (12%) than adult (7.8%) patients. However, this should be interpreted cautiously, because this was not a national survey and the majority were preselected surgical patients.
Diagnosis
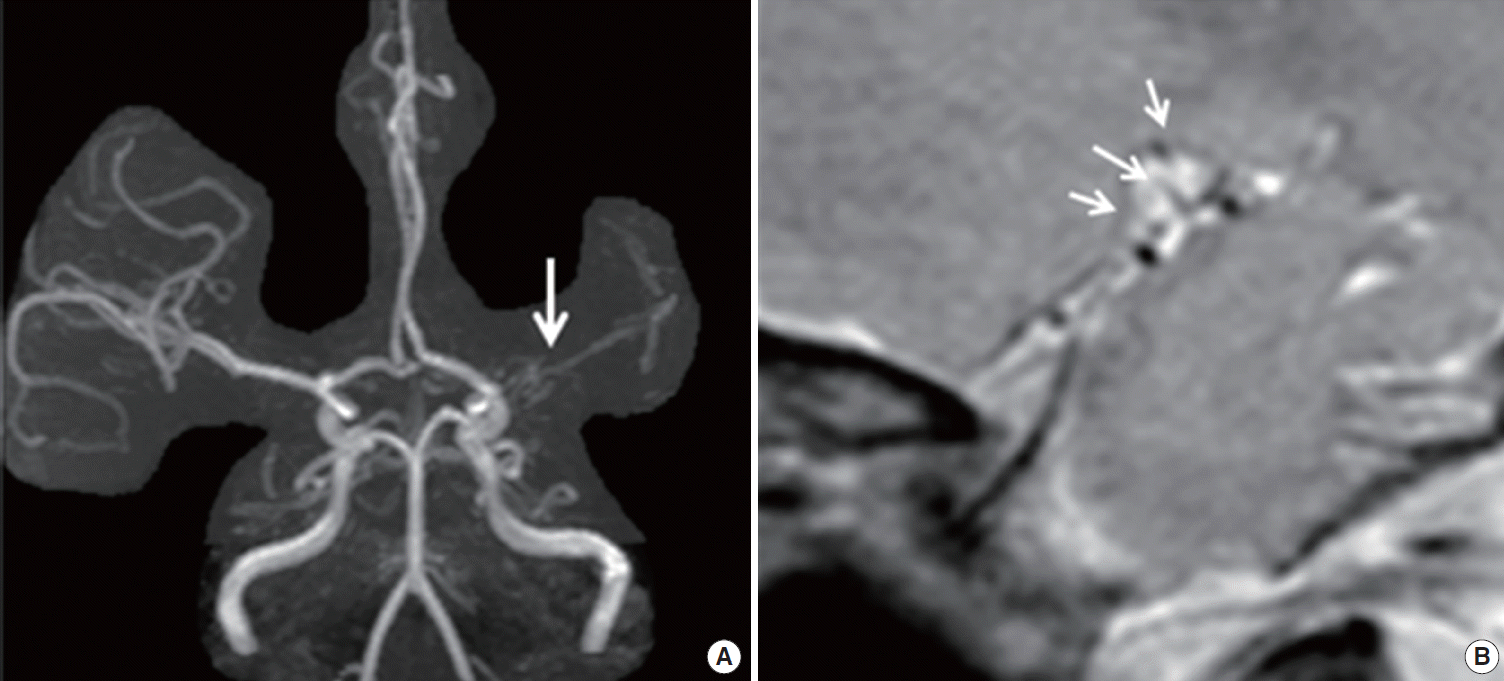
Diagnostic criteria previously required that steno-occlusive change at the ICA should be evident bilaterally for the definitive diagnosis of MMD. Recently, however, diagnostic criteria for definitive MMD were revised to include patients with unilateral terminal ICA steno-occlusion as well. Definitive diagnosis of MMD requires catheter angiography in unilateral cases, while bilateral cases can be promptly diagnosed by either catheter angiography or MRA (Figures 2 and 3). Although catheter angiography is the most important diagnostic test, this relatively invasive technique is associated with procedure-related complications, and therefore preprocedural measures including sufficient hydration should be appropriately taken, especially in pediatric patients. Based on various angiographic findings, Suzuki and Takaku [1] proposed 6 stages of angiographic evolution. However, a stepwise progression from stage 1 through stage 6 has been observed in only a limited number of patients [42], and the practical value of the classification remains unclear. MRA and computed tomography angiography are noninvasive tests that can detect distal ICA or MCA steno-occlusion. However, they have limited sensitivity in detecting basal collaterals. Modern supportive diagnostic tools are useful in making an accurate diagnosis of MMD, especially in patients in an early stage of Suzuki’s angiographic grading system [1], when an abnormal vascular network is not yet evident. To improve diagnostic capability, high resolution vessel wall MRI has recently been used. Kaku and colleagues proposed a constrictive remodeling theory in which outer diameter narrowing of the affected intracranial vessels was an early characteristic change of MMD, as demonstrated by three-dimensional (3D) constructive interference in steady-state (CIISS) MRI [48]. Yuan et al. also reported that vascular wall thinning and arterial outer diameter narrowing shown by high resolution MRI could be early morphological changes characteristic of MMD [49]. Besides outer diameter narrowing, concentric enhancement of symptomatic segments was observed in MMD patients [50]. Thus, high-resolution MRI including 3D-CIISS could provide supportive information for the accurate diagnosis of MMD, especially in the early angiographic stage. In addition, genetic analysis as described in another review would provide supportive information for the diagnosis of MMD (Figure 5).
Summary
Recent epidemiological studies have confirmed that MMD is most common in East Asian countries such as Korea and Japan. Although the prevalence of MMD appears to be slightly lower among the Chinese, larger national epidemiological studies are still needed in China. In East Asian countries, clinical features of MMD are similar; there are two peaks of incidence, at around 10 years and at 30-40 years, with different clinical presentations. In children, ischemic symptoms, especially TIA, are predominant, whereas intracranial hemorrhage is more common in adult patients. In Asian countries, the incidence of MMD appears to increase over time. Patients with MMD are less frequently described in the West, and there seems to be a lower incidence of familial cases. These differences may be attributed to the more significant role of MMD genetic susceptibility in East Asians than in Caucasians. However, reliable studies are still needed to understand the epidemiology, pathophysiology, and genetics of MMD.